
![]()
DEET
(N,N-Diethyl-meta-toluamide)
The spray-on insect repellant
![]()
Paul May
University of Bristol
![]()
Molecule of the Month January 2011
Also available: JSMol version.
![]()

 |
DEET(N,N-Diethyl-meta-toluamide)The spray-on insect repellant
Paul May
Molecule of the Month January 2011
|
 |
 Why make an insect repellant?
Why make an insect repellant?Insect bites and stings, as well as being painful and annoying, can be lethal. Mosquito bites, in particular, transmit malaria to humans, and it is estimated to cause approximately 3 million deaths every year worldwide. Insect bites and stings can also cause allergic reactions in people which can sometimes be fatal. So a chemical spray which can ward off insects is a potentially life-saving invention.
Over the years various types of substances have been used to repel mosquitos. These include such things as smoke, plant extracts, oils, tars, and muds. However, the first really effective ingredient used in mosquito repellents was citronella oil - a herbal extract derived from the citronella plant. The insect repelling properties of citronella oil was only discovered by accident in 1901 when it was used as a hairdressing fragrance. It is believed that the chemical terpenes of which it is composed are responsible for its repellent activity. Unfortunately, citronella oil is very volatile and evaporates too quickly from surfaces to which it is applied. Also, large amounts are needed to be effective.
These disadvantages inspired researchers to search for more effective alternative compounds. Many of the early attempts at creating synthetic insect repellents were initiated by the United States military. Out of this research came the discovery of the repellent dimethyl phthalate in 1929. This material showed a reasonable level of effectiveness against certain insect species, but it was ineffective against others. Two other materials were developed as insect repellents: Indalone was found to repel insects in 1937, and Rutgers 612 (2-ethyl-1,3-hexane diol) was synthesized soon after. Like dimethyl phthalate, these materials had certain limitations which prevented their widespread use.
In 1955, scientists synthesized DEET (N-N-diethyl-meta-toluamide), which is currently the most widely used active ingredient for mosquito repellents, and is found in creams, lotions, and aerosols.
|
Most repellent chemicals are thought to work work by interfering with the mosquito's homing system. A mosquito's antennae contain a number of chemical receptors which are sensitive to different compounds, such as CO2 (exhaled by animals) and lactic acid, which naturally evaporates from the skin of warm-blooded animals. The receptors allow the mosquitos to follow these emissions back to their source, and hence 'home in' on their prey [Ditzen et al, 2008]. However, when a repellent is applied to the skin it evaporates and masks the scent of the lactic acid. Thus, the mosquito is effectively 'blinded'. More recent research has shown that mosquitos, as well as being 'blinded' by the chemical, actively dislike the smell of DEET [Syed and Leal, 2008].
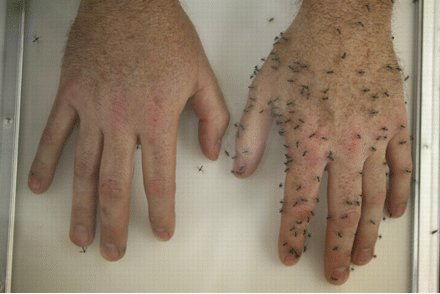
Guess which hand has had DEET applied to it!
DEET is a slightly yellow liquid at room temperature. It is prepared by acylating meta-toluic acid, and then reacting this with diethylamine.
 How much is needed?
How much is needed?DEET is usually applied to the skin, but the active ingredient must evaporate from the surface to work, the repellent activity lasts for a limited time, depending upon the concentration used. 100% DEET was found to offer up to 12 hours of protection while several lower concentration DEET formulations (20%-34%) offered 3–6 hours of protection. The Center for Disease Control recommends 30-50% DEET to prevent the spread of pathogens carried by insects.
Products that use DEET or citronella oil as the primary active ingredients have been reported to causes rashes in some people, and there have been increasing debates as to whether DEET is a safe substance to use on human skin. DEET has has extensive independent research into its safety for decades and has proved safe to use as long as usage instructions are followed.
Now that scientists are beginning to understand how molecules interact with the mosquito's receptors, it is becoming possible to design newer, more effective repellent molecules by modifying their structure [Katritzky et al, 2008]. N-acylpiperidines are a group of molecules that show great promise in this area, and the goal is that by tailoring the chemical structure of these molecules an insect repellent can be developed that is safe, non-toxic, and 100% effective.
![]()
![]()
![]() Back to Molecule of the Month page. [DOI:10.6084/m9.figshare.5255203]
Back to Molecule of the Month page. [DOI:10.6084/m9.figshare.5255203]