

Molecule of the Month - February 2002
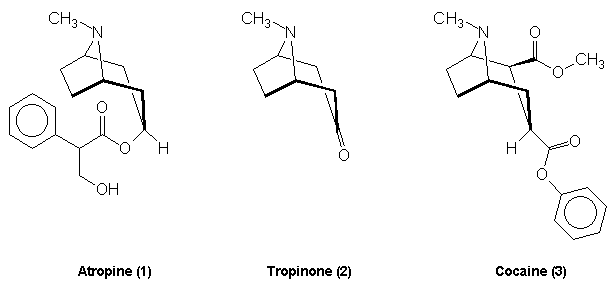
Atropine is a white solid with melting point 118oC. It is a monocyclic alkaloid which has the molecular formula C17H23O3N. Its structure is shown below:
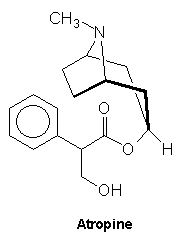
It is a highly toxic material which causes fatalities when ingested in even moderate quantities. It is used in medicine as a muscle relaxant and finds application as an anti-spasmodic which may be used as a pretreatment before abdominal surgery and optometry to dilate the pupils, where it relaxes the muscles of the eye.
It is quite difficult to give a precise definition for the class of compounds referred to as alkaloids [1]. However it is probably fair to say that the definition proposed by Ladenburg in the late 1880s is probably reasonable and still valid today. Ladenburg suggested that alkaloids were compounds:
derived from plants
with a basic character (hence the term alkaloid from alkali)
contained a nitrogen based heterocylic ring within their molecules
In general alkaloids are physiologically very active and often extremely poisonous - they are the "bad guys" of the natural products world. Having said this, many find applications in medicine when administered in small doses.
Alkaloids can be sub-categorised [2] according to:
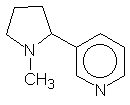
Monocyclic alkaloids - which contain only one ring. A typical monocyclic alkaloid is nicotine:
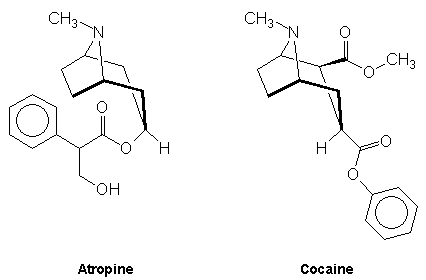
Bicyclic alkaloids - these may be illustrated by the tropane alkaloids which consist of molecules with a 1,4 nitrogen bridged cycloheptane structure. Atropine and cocaine are examples
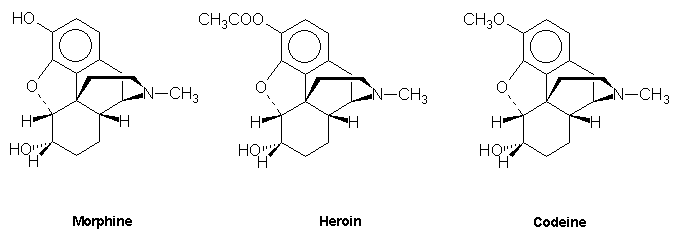
Polycyclic alkaloids such as strychnine, lysergic acid, cannabinol, morphine, heroin and codeine.
What are neurotoxins?
These are poisons, which include atropine, various other alkaloids, some snake and spider venoms and nerve gas, which disrupt the workings of the nervous system.
What is the nervous system?
This consists of billions of nerve cells or neurones [3]. Neurones are special elongated cells which can carry electrical impulses along their length. The nervous system is divided into two sections:
Central Nervous System (CNS) which consists of the brain and spinal cord. The CNS co-ordinates and integrates information it receives and organises responses
Peripheral Nervous System (PNS) which consists of nerves running from the CNS to the peripheries - arms, legs, abdomen, etc. The role of the PNS is to collect information about changes in the environment registered by receptors and pass this on to the CNS and to pass impulses from the CNS to the effectors, for example skeletal muscles, which respond accordingly.
Neurones themselves fall into three classes:
Motor neurones are nerve cells which transmit impulses from the CNS to the effectors, for example muscles.
Sensory neurones are nerve cells which transmit impulses from the receptors to the CNS.
Intermediate neurones which are nerve cells found in the CNS and which connect sensor and motor neurones with each other and with other cells in the CNS
Neurones can be very long, perhaps up to a metre in length and impulses travel along them from one end to the other at speeds of up to 100 m s-1 (over 220 m.p.h.!)
How does the Impulse pass along the neurone?
The long, cylindrical section of the neurone along which the impulse passes is known as the axon. This membrane of the axon does not conduct electricity and is normally impervious to the passage of ions such as Na+, and K+. However it is possible to force cations through this membrane by an energy absorbing "sodium pump". The system so arranges itself so that the majority of sodium ions reside outside the membrane and most of the potassium ions lie within the cell plasma. The numbers of sodium and potassium ions transferred is not equal and this results in an electrical potential of about 60 mV being set up across the membrane, the inside being negative and the outside being positive. This potential remains constant while the axon is at rest and so is referred to as the resting potential.
When an impulse passes down the axon the situation changes for an instant. As the impulse reaches a particular point on the axon the membrane becomes more permeable to sodium ions. The sodium ions, being in higher concentration on the outside of the membrane than in the plasma, rush into the interior of the cell and carry their charge with them. This suddenly reverses the potential and the interior becomes positive for an instant. This is known as the action potential. After this, the potassium ions rapidly migrate through the membrane to the outside, so reducing the positive charge within the cell and re-establishing the resting potential. The sodium and potassium pumps then re-establish the original concentrations of cations so that the process may be repeated. As the process occurs at one point in the axon, so it initiates a similar process at an adjacent site and so the impulse propagates along the axon in a wave like motion.
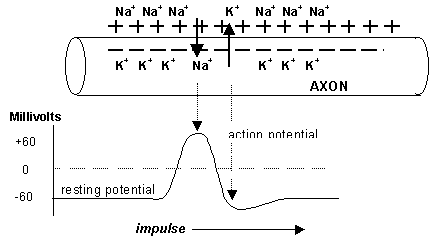
So what happens when the impulse reaches the end of the axon?
The point at which neurones meet is called a synapse. The end of an axon is a somewhat bulbous region known as a synaptic knob. The synaptic knob is full of mitochondria and small sac-like vesicles containing neurotransmitters such as acetylcholine and it is sealed by a membrane. There is a small gap approximately 20 nm wide, between this membrane and that of the next cell and this is known as the synaptic cleft. The membrane of the cell through which the impulse is being transmitted is called the pre-synaptic membrane while that of the cell to which the signal is to be transferred is called the post-synaptic membrane.
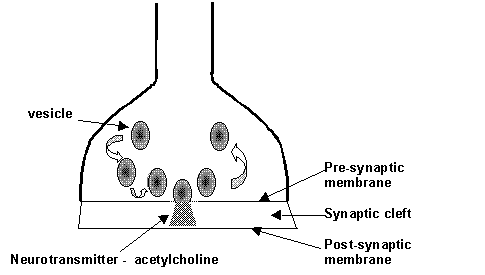
When the impulse reaches the synaptic knob it initiates an inflow of Ca2+ ions which causes the vesicles to move towards, and become attached to, the pre-synaptic membrane. The vesicles then discharge their neurotransmitter substance (acetylcholine) into the synaptic cleft. This material rapidly diffuses to the post-synaptic membrane where it initiates a process which generates an impulse in this cell. If the neurotransmitter remained bound to the site on the post-synaptic membrane it would maintain constant stimulation of the cell resulting in the repeated generations of impulses in the cell. The cell would become fatigued and cease to transmit pulses and so would become ineffective. To prevent this situation from occurring the acetylcholine receptor has an enzyme attached to it which rapidly decomposes the neurotransmitter once it has initiated a response. The enzyme acting on acetylcholine is called cholinesterase and it converts acetylcholine into ethanoic (acetic) acid and choline. The choline then diffuses back to the pre-synaptic membrane where it is reabsorbed and converted back into acetylcholine in the vesicles. If this enzyme ceased to act, not only would the post-synaptic cell become inactive due to constant stimulation resulting in fatigue, but the store of acetylcholine in the pre-synaptic cell would rapidly become exhausted and this cell too would become inactive.
…And so to neurotoxins
The chemical nature of synaptic transfer makes them very susceptible to disruption by toxins. Many alkaloid poisons, snake and spider venoms and nerve gases act in this way [4], [5].
Atropine acts by preventing acetylcholine from depolarising the post-synaptic membrane and so prevents generation of the impulse in this cell. Curare has a similar effect but acts at the junction between nerve cells and muscles.
a-Bungarotoxin, from the venom of snakes of the genus Bungarus operate in a similar fashion to curare. Eserine prevents cholinesterase from catalysing the hydrolysis of acetylcholine, thus stimulating the post-synaptic cell to generate a constant series of impulses until it is fatigued. Strychnine enhances synaptic transmissions to such an extent that the slightest stimulation will cause convulsive contractions in the victim's muscles. Some nerve gases used for military purposes act in the same way. a-Latrotoxin, a protein produced by the black widow spider induces massive acetylcholine release while Botulinus Toxin, consisting or a mixture of eight proteins produced by Clostridium botulinum, inhibits the release of acetylcholine at the neuromuscular junction.Source
Atropine occurs in the deadly nightshade plant (Atropa belladonna) alongwith hyoscyamine. This plant has brown-purple flowers and berries which change from red to purple as the summer progresses

It is often confused with the much less toxic woody nightshade or bittersweet (Solanum dulcamara) which has flowers with purple petals and a yellow centre

Structure
Atropine
The structure of atropine was investigated by R Willstätter in the late 1890's. It was found that on hydrolysis, atropine gave (±)-tropic acid and tropine, which was shown to be an alcohol.
Tropic acid was shown to have a molecular formula of C9H10O3 and lost a molecule of water to yield atropic acid on strong heating. Atropic acid, C9H8O2, had the structure (1) implying that tropic acid was either: compound (2) or (3):
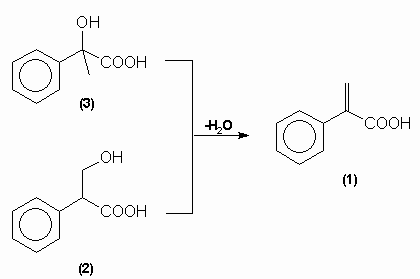
Mackenzie and Wood showed that tropic acid was compound (2), 3-hydroxy-2-phenylpropanoic acid, by unambiguous synthesis in 1919 while Fodor, et al, demonstrated the absolute configuration by its correlation with (-)-alanine in 1961:
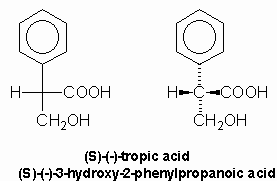
Tropine, C8H15ON contains a hydroxyl group and behaves as a saturated compound. Ladenburg (1883, 1887) demonstrated that the molecule contained a reduced pyridine nucleus. His work lead him to propose two possible structures for tropine, (4) and (5):
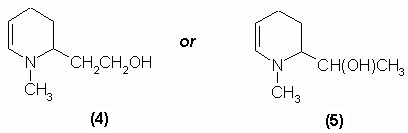
Merling (1891) obtained tropinic acid by oxidation of tropine using chromium trioxide. Tropinic acid was shown to be a dicarboxylic acid which contained the same number of carbon atoms as tropine:
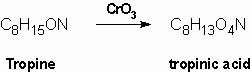
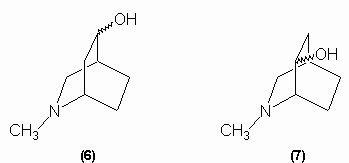
Since tropinic acid is a dicarboxylic acid and no carbon is lost in the oxidation, the alcohol group oxidation must have involved ring cleavage and so the hydroxyl group had to be within a ring. This made the Ladenburg structures untenable and so Merlin proposed either structure (6) or (7) for tropine:
Willstätter (1895 - 1891) notes that tropinone was formed during oxidation prior to ring cleavage. This substance was a ketone which was demonstrated to contain the CH2COCH2moiety by the formation of dibenzylidene and di-oximino derivatives. This, in turn, made Merling's structures untenable and so Willstätter proposed structure (8) for tropine:
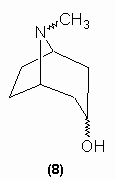
The structure of tropine has been confirmed to be (8) by synthesis by Willstätter (1900 - 1903) and by Robinson (1917) - see section on synthesis.
Synthesis
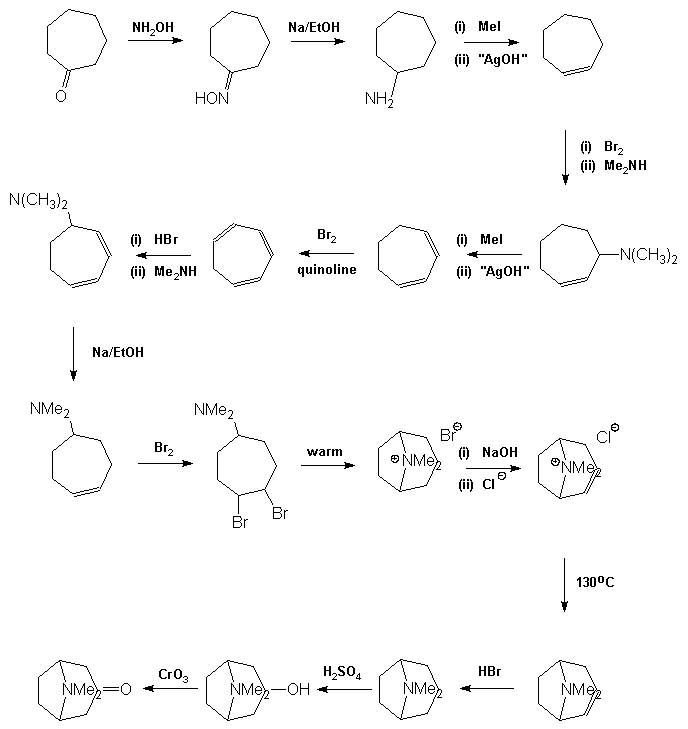
Tropinone The first synthesis of atropine was achieved by Richard Willstätter in 1901 [7]. Before continuing
the discussion of his work it is perhaps useful to remind ourselves of the situation which faced organic chemists at this time. Recent innovations
such as mass spectrometry, infrared spectroscopy and nuclear magnetic resonance spectroscopy which are taken for granted today, were simply not
available to assist in the determination of structure. Instead chemists had to rely on hard won information based upon simple chemical tests. This
information was often inadequate and incomplete and the determination of structure was a process of detective work which often required great
intuition and creativity. At this time structure determination was not always equivocal and final proof could only be established by unambiguous
synthesis of the compound with the suspected structure followed by comparison with an authentic sample of the natural product. Thus synthesis
was often a matter of utilitarian necessity rather than the creative, elegant art form illustrated by the work of many of the great synthetic
chemists such as Woodward and Corey. Richard Willstätter was a giant in the field of plant natural products and
this was recognised when he was awarded the Nobel Prize for Chemistry in 1915 [8, click on image to link to Nobel site]. He was interested in many alkaloids including atropine (1) and the structurally similar cocaine (3) molecule. Both of these compounds yielded tropinone (2) as a degradation product
and so his synthesis targeted this molecule initially. It was relatively straightforward to reduce tropinone to tropine and hence form its ester
atropine. His preparation of tropinone is a competent but long synthesis which demonstrates one of the
fundamental difficulties involved in the preparation of complex organic molecules. Although the individual steps in the synthesis generally give
good to excellent yields, there are many of them which means that the overall yield becomes diminishingly small, of the order of 1%. As a
result the early steps in the synthesis have to be carried out on inconveniently large quantities of material, and despite this,
usually have to be repeated several times in order to obtain sufficient material to carry out the later stages on an acceptable scale. In 1917 Robinson [9] approached the synthesis in a totally radical way. In his own words: © The Nobel Foundation "By imaginary hydrolysis at the points indicated by the dotted
lines, the substance may be resolved into succinaldehyde methylamine and acetone. ...It
was proved that tropinone is obtained in small yield by condensation of succinaldehyde with acetone and methylamine in aqueous solution. An improvement followed by the replacement of acetone by a salt [calcium] of acetone dicarboxylic acid. The initial product is a salt of tropinone dicarboxylic acid, and this loses two molecules of carbon dioxide with the formation of tropinone when the solution is acidified and heated" [10, click on image to link to
Nobel site]. This resulted in the formation of tropinone in one step. More recent work
by Schöpf has allowed the yield for this reaction to be raised to about 90%, mainly by carrying out the processed under buffered conditions. For a more detailed account of these syntheses it is worth reading the account by Ian
Fleming in his book Selected Organic Syntheses, A Guidebook for Organic Chemists [11]. Tropinone may then be converted into tropine by metal in acid reduction, the best
yields being obtained using zinc in HI. It may be noticed that the final precursor in the Willstätter synthesis appears to
be tropine. This is not the case as the material is its geometric isomer, j -tropine,
and thus tropine is formed by oxidation of j
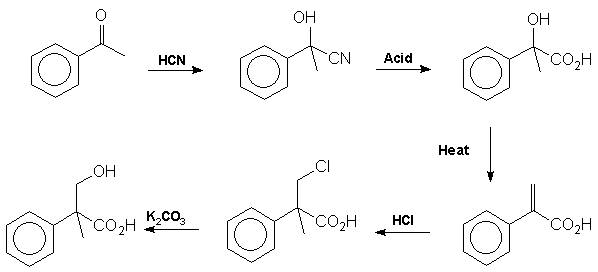
-tropine to tropinone followed by stereoselective reduction of the carbonyl group. Tropic acid Mackenzie and Ward [12] proved the structure of tropic acid by synthesis from
acetophenone in 1919: Note that the addition of HCl in step 4 contravenes Markownikoff's rule.
This is presumably due to the electron withdrawing effect of the carboxyl group which destabilises the tertiary carbonium ion intermediate relative
to the primary carbonium ion. It is tropic acid which introduces the stereocentre into the atropine molecule. The racemic mixture formed
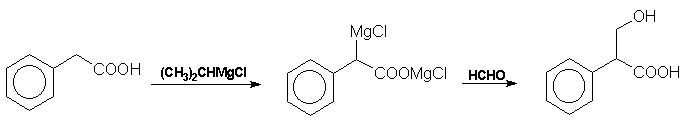
from this reaction sequence may be resolved by reaction with quinine followed by fractional crystallisation of the diastereoisomers.. More recently Blicke et al have prepared tropic acid from phenylacetic
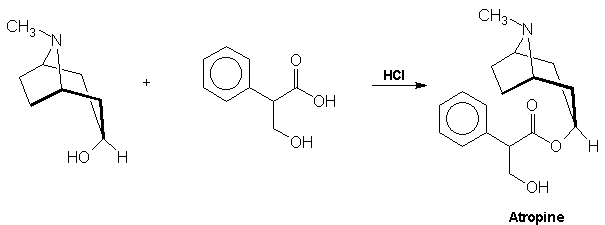
acid via a Grignard reagent and formaldehyde: Atropine The final problem in the synthesis, the combination of tropine and
tropic acid, was overcome by a Fischer-Speier esterification [13].
The acid and alcohol were heated together in the presence of HCl to yield atropine References
1
Finar,
I.L., Organic Chemistry Vol. 2 4th. Edn., Longman 1965, p.
601
2
http://www.staffs.ac.uk/schools/sciences/chemistry/tebby/alkaloids.html
3
Roberts, M. B. V., Biology, A Functional Approach 2nd. Edn.,
Thomas Nelson and Sons 1979, pps. 262 et seq
4
http://web.indstate.edu/thcme/mwking/nerves.html#synaptic
5
http://www.pharmcentral.com/neurotransmitters.htm#1-neurotransmitters
6
Finar, I.L., Organic Chemistry Vol. 2 4th. Edn., Longman 1965,
pps. 631 - 639
7
Willstätter, R., Ber., 34, (1901), pp.
129 and 3163
8
http://www.nobel.se/chemistry/laureates/1915/
9
Robinson, R., J. Chem. Soc.,
(1917), p762 10
http://www.nobel.se/chemistry/laureates/1947/index.html 11
Fleming, I., Selected Organic Syntheses, John Wiley
1973, pps. 17 - 23 12
Finar, I.L., Organic Chemistry Vol. 2 4th. Edn., Longman 1965,
pps. 631 et seq.
13
Finar, I.L., Organic Chemistry Vol. 2 4th. Edn.,
Longman 1965, p. 636
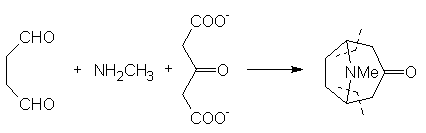
![]() Back to Molecule of the Month page. [DOI:10.6084/m9.figshare.5450335]
Back to Molecule of the Month page. [DOI:10.6084/m9.figshare.5450335]