
The discovery and characterisation of the structure of ferrocene, Fe(C5H5)2 in the early 1950's, led to an explosion of interest in d-block metal carbon bonds and brought about development and the now flourishing study of organometallic chemistry.
Prior to the 1950's few d-block organometallics were synthesized and characterised. The first (1), an ethylene complex of platinum(II), was prepared by W.C. Zeise in 1827. In 1890's Ludwig Mond and co-workers synthesised the first metal carbonyl, tetracarbonynickel (2). However, the structures of such complexes were difficult to deduce using chemical methods, and thus it wasn't until the 1950's when NMR and single crystal X-ray diffraction could be used to solve the structures of these complexes in solution and solid state respectively.
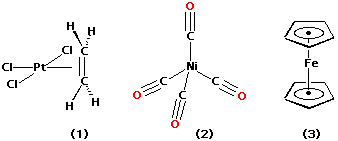
In 1951, ferrocene (3), was first prepared. It was found to be unusually
stable, and its structure and bonding defied conventional bonding descriptions.
The sandwich structure of ferrocene was first predicted from its IR and
NMR and then confirmed by X-ray crystallography by 1954.
The rapid growth in the study of organometallic compounds by research groups around the world led to the Nobel Prize awarded in 1974 to Ernst Fisher and Geoffrey Wilkinson1 for their contribution to the field.