GalCer as receptor for pathogens
Glycosphingolipids like GalCer are used
as cellular binding sites for a wide variety of pathogens, including viruses,
bacteria, fungi and parasites (11). Namely,
GalCer is recognized by the following: HIV-1, prions, Borrelia burgdorferi (the
causative agent of Lyme disease; Borrelia burgdorferi bacteria
are transmitted to humans by the bite of infected deer ticks and caused more
than 23,000 infections in the
In addition to providing a wide variety
of accessible sugar motifs, glycosphingolipids in lipid rafts are endocyted
through a specific pathway. Indeed, internalization of ligands bound to lipid
rafts is translocated to various cellular compartments (endoplasmic reticulum,
nucleus, apical or basolateral membrane) but not to lysosomes. Thus, by chosing
lipid rafts as a pathway of entry, microbes avoid potential lysosomal
degradation. In other words, lipid rafts may be viewed as membrane-organelles
shuttles that take the pathogens aboard on the cell surface and drive them to a
protected intracellular niche (16).
Involvement
of GalCer in HIV-1 enteropathy.
Malabsorption and diarrhea are common,
serious problems in AIDS patients, and are in part due to the incompletely
understood entity HIV enteropathy. The HIV-1 surface envelope glycoprotein
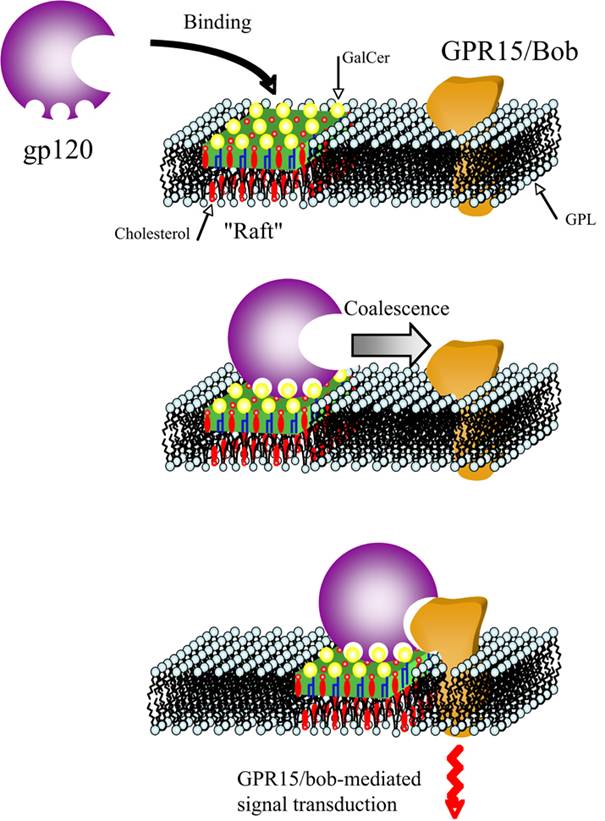
gp120 binds to GalCer on the enterocyte
cell surface. Then, the complex is delivered to a membrane protein called
GPR-15/Bob. GalCer-contaninig lipid rafts allow the migration of HIV-1 on the
cell surface until reaching GPR15/Bob. GPR15/Bob is a receptor with seven
transmembrane domains coupled to G proteins. Thus, gp120-induced stimulation of
this receptor results in a cascade of activation leading to a burst of
intracellular calcium concentration, extensive microtubule depolymerization, an
80% decrease in intestinal barrier function and, and a 70% decrease in
intestinal glucose absorption. These changes are also evidenced in the
intestinal epithelium of AIDS patients suffering of enteropathy. Antibodies
against GalCer (as well as anti-GPR15/Bob antibodies) block this suite of
events and efficiently protect the cells from HIV-1 injury. As HIV-1 does not
need to infect the intestinal cells to impair intestinal function, HIV-1
gp120 can be considered as a ‘virotoxin’
(17-19).