  |
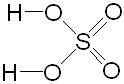
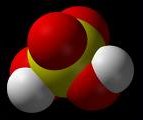
SULFURIC ACID
Mike Thompson
Also available: HTML, Chime Enhanced and JMol versions.
|
  |
  |
SULFURIC ACID
Mike Thompson
Also available: HTML, Chime Enhanced and JMol versions.
|
  |
 Spelling
SpellingIf you wish this molecule of the month was on sulphuric acid rather than sulfuric acid then please click on the danger sign (right). It used to be known as 'oil of vitriol'. In fact it might be even better to call it sulfur(VI) acid.
 Contact Process
Contact ProcessSulfuric acid is produced in larger quantities than any other acid. Almost every manufactured item in the modern world comes into contact with H2SO4 at some stage in its history. It is so important, that at one time the annual production of sulfuric acid was taken as a measure of the degree of industrialisation of a country, and earned it its nickname of the 'king of chemicals'.
Sulfuric acid (H2SO4) is still an extremely important chemical used in the manufacture of fertilisers and explosives. It is made nowadays from sulfur dioxide by the Contact Process. This process reacts together oxygen, O2 and sulfur dioxide, SO2 at 450°C and 2 atmospheres pressure, with the help of vanadium(V) oxide, to make sulfur trioxide, SO3 gas in a 98% yield. This is then dissolved in water to make H2SO4. [Note that directly dissolving SO3 in water is not practical due to the highly exothermic nature of the reaction, forming a corrosive mist instead of a liquid. Instead, SO3 can be absorbed into existing H2SO4 to produce oleum (H2S2O7), which may then be mixed with water to form sulfuric acid]. The original reaction to make SO3 has a high activation energy and without the catalyst is therefore very slow. It only became economically viable when in 1746, John Roebuck developed the lead chamber process, which catalysed the reaction using oxides of nitrogen through the intermediate formation of HOSO2ONO. It could be argued that the discovery of this process catalysed the Industrial Revolution (as well as the reaction!) in the UK, and as a result even helped to forge the British Empire!
At room temperature pure sulfuric acid (100%) is a covalent liquid. Concentrated sulfuric acid is 98% (18.7M) and is an oily liquid with a density of 1.83 g/cm3. It decomposes at its b.p. (330°C) and forms white fumes.
H2SO4 (l) ![]() H2O (g) + SO3 (g).
H2O (g) + SO3 (g).
The reverse reaction is how to make sulfuric acid.
For safety data, click here.
It has several important properties when concentrated; acid, oxidizing agent and dehydrating agent which makes it a very versatile reagent. It tastes sour not that you would taste it in a laboratory! It turns blue litmus red except when it is the covalent molecule, i.e. 100% H2SO4.
Concentrated sulfuric acid is very hygroscopic. Its affinity for water is so great that if you left a beaker half full with conc. H2SO4 for three weeks, its volume would greatly increase and it would become more dilute because it would have absorbed so much water vapour out of the air.
Firstly, add dilute HNO3. This is to prevent precipitation of other insoluble barium compounds such as BaCO3 or BaSO3. Secondly, add Ba(NO3)2 (aq). If sulfuric acid or a sulfate is present a white precipitate will be immediately observed.

![]()

Ba2+ (aq) + SO42- (aq) ![]() BaSO4 (s) [White]
BaSO4 (s) [White]
Hot concentrated sulfuric acid is an oxidising agent when it accepts electrons. The electrons are supplied by the reducing agent in the reaction:
2 H2SO4 (aq) + 2e- ![]() SO42- (aq) + 2 H2O (l) + SO2 (g)
SO42- (aq) + 2 H2O (l) + SO2 (g)
Zn (s) + 2 H2SO4 (conc) ![]() ZnSO4 (aq) + 2H2O (l) + SO2 (g)
ZnSO4 (aq) + 2H2O (l) + SO2 (g)
Fe (s) + 2 H2SO4 (conc) ![]() FeSO4 (aq) + 2H2O (l) + SO2 (g)
FeSO4 (aq) + 2H2O (l) + SO2 (g)
Mg (s) + 2 H2SO4 (conc) ![]() MgSO4 (aq) + 2H2O (l) + SO2 (g)
MgSO4 (aq) + 2H2O (l) + SO2 (g)
Cu (s) + 2 H2SO4 (conc) ![]() CuSO4 (aq) + 2H2O (l) + SO2 (g)
CuSO4 (aq) + 2H2O (l) + SO2 (g)
S (s) + 2 H2SO4 (conc) ![]() 2 H2O (l) + 3 SO2 (g)
2 H2O (l) + 3 SO2 (g)
C (s) + 2 H2SO4 (conc) ![]() 2 H2O (l) + 2 SO2 (g) + CO2 (g)
2 H2O (l) + 2 SO2 (g) + CO2 (g)
Sulfur dioxide can be detected with a strip of filter paper moistened with potassium dichromate which turns from orange to green.
Chlorides only react in one way, no matter what the temperature.
NaCl (s) + H2SO4 (l) ![]() HCl (g) + NaHSO4 (s)
HCl (g) + NaHSO4 (s)
The misty fumes of hydrogen chloride gas are seen. The reason chlorine gas is not formed is because sulfuric acid is not powerful enough to oxidise chloride ions.
Bromide ions are a stronger reducing agent than chloride ions. As bromide ions are larger than chloride ions their electrons are easier to remove. Bromide ions reduce sulfuric acid into sulfur dioxide in two steps.
NaBr (s) + H2SO4 (l) ![]() HBr (g) + NaHSO4 (s)
HBr (g) + NaHSO4 (s)
2 HBr (g) + H2SO4 (l) ![]() Br2 (l) + 2 H2O (l) + SO2 (g)
Br2 (l) + 2 H2O (l) + SO2 (g)
Iodide ions are even larger than bromide ions and iodides electrons are even easier to remove. Iodide ions will reduce concentrated sulfuric acid into hydrogen iodide gas, which will further reduce sulfuric acid in three ways at room temperature.
NaI (s) + H2SO4 (l) Equation 1: 8 HI (g) + H2SO4 (l) ![]() 4 I2 (s) + H2S (g) + 4 H2O (l)
4 I2 (s) + H2S (g) + 4 H2O (l)
Equation 2: 6 HI (g) + H2SO4 (l) ![]() 3 I2 (s) + S (s) + 3 H2O (l)
3 I2 (s) + S (s) + 3 H2O (l)
Equation 3: 2 HI (g) + H2SO4 (l) ![]() I2 (s) + SO2 (g) + H2O (l)
I2 (s) + SO2 (g) + H2O (l)
What might you expect to observe if conc. sulfuric acid was added to solid sodium iodide?
 Uses of Sulfuric acid
Uses of Sulfuric acid superphosphate manufacture
superphosphate manufacture As a dehydrating agent
As a dehydrating agentConcentrated sulfuric acid is very good at removing the water from sugars. When the elements of water are removed from sucrose it leads to a black mass of carbon (see photo, left and the Molecule of the Month page on glucose).
C12H22O11 (s) + n H2SO4 (l) ![]() 12 C (s) + 11 H2O (l) + n H2SO4 (l)
12 C (s) + 11 H2O (l) + n H2SO4 (l)
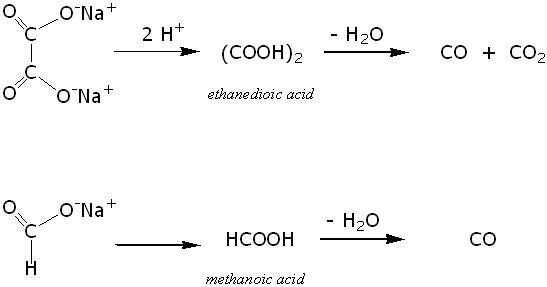
Sulfuric acid can also be used to dehydrate sodium salts of some carboxylic acids. This use of sulfuric acid has already been mentioned in the Molecule of the Month page on Carbon monoxide. The reactions involve protonation by the acid followed by subsequent dehydration.
Dilute sulfuric acid neutralises basic oxides or hydroxides to form sulfate salts and water. Don't forget sulfuric acid is a strong acid (completely ionizes in water).
CuO (s) + H2SO4 (aq) ![]() CuSO4 (aq) + H2O (l)
CuSO4 (aq) + H2O (l)
ZnO (s) + H2SO4 (aq) ![]() ZnSO4 (aq) + H2O (l)
ZnSO4 (aq) + H2O (l)
Cu(OH)2 (s) + H2SO4 (aq) ![]() CuSO4 (aq) + 2 H2O (l)
CuSO4 (aq) + 2 H2O (l)
Zn(OH)2 (s) + H2SO4 (aq) ![]() ZnSO4 (aq) + 2 H2O (l)
ZnSO4 (aq) + 2 H2O (l)
As sulfuric acid is dibasic (ionizes in two stages) when it reacts with sodium hydroxide it can form two possible sodium salts. To see which one is favoured you need to consider the stoichiometry (number of moles in the balanced equation) for the following two reactions.
2 NaOH (aq) + H2SO4 (aq) ![]() Na2SO4 (aq) + 2 H2O (l)
Na2SO4 (aq) + 2 H2O (l)
NaOH (aq) + H2SO4 (aq) ![]() NaHSO4 (aq) + H2O (l)
NaHSO4 (aq) + H2O (l)
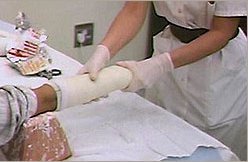
 Dilute sulfuric acid neutralises basic carbonates to form sulfate salts, water and carbon dioxide, resulting in effervescence (as shown in the photo, right).
Dilute sulfuric acid neutralises basic carbonates to form sulfate salts, water and carbon dioxide, resulting in effervescence (as shown in the photo, right).
Na2CO3 (aq) + H2SO4 (aq) ![]() Na2SO4 (aq) + H2O (l) + CO2 (g)
Na2SO4 (aq) + H2O (l) + CO2 (g)
MgCO3 (aq) + H2SO4 (aq) ![]() MgSO4 (aq) + H2O (l) + CO2 (g)
MgSO4 (aq) + H2O (l) + CO2 (g)
The rate of reaction not only depends on the surface area of the carbonate and the concentration and temperature of the acid, but also on the solubility of the sulfate. When marble (CaCO3) reacts with dilute H2SO4 it initially effervesces but because calcium sulfate is only sparingly soluble in water, once it forms as a deposit on the marble surface, the reaction soon slows.
Dilute sulfuric acid reacts with metals higher than hydrogen in the reactivity series to form sulfate salts and hydrogen gas. Note the metal reactions are different with concentrated sulfuric acid.
Mg (s) + H2SO4 (aq) ![]() MgSO4 (aq) + H2 (g)
MgSO4 (aq) + H2 (g)
Zn (s) + H2SO4 (aq) ![]() ZnSO4 (aq) + H2 (g)
ZnSO4 (aq) + H2 (g)
Fe (s) + H2SO4 (aq) ![]() FeSO4 (aq) + H2 (g)
FeSO4 (aq) + H2 (g)
Iron is cleaned free from rust prior to coating with tin to form tinplate and with zinc to form galvanized iron.
 The biggest (80%) use of sulfuric acid in the manufacture of sulfates.
The biggest (80%) use of sulfuric acid in the manufacture of sulfates.
Gypsum is CaSO4.2H2O and is used to make plaster board as well as plaster casts (see photo, right). The laxative 'epsom salt' is MgSO4.7H2O, and green vitriol is FeSO4.7H2O. In 1648, Glauber made sulfuric acid by distilling crystals of this salt. Potassium sulfate is unusual in that it crystallizes without water of crystallization and has the formula K2SO4. As sulfuric acid is dibasic, (diprotic) it is capable of forming two salts depending on the amount of alkali present in the reaction mixture;
2 NaOH (aq) + H2SO4 (aq) ![]() Na2SO4 (aq) + H2O (l)
Na2SO4 (aq) + H2O (l)
NaOH (aq) + H2SO4 (aq) ![]() NaHSO4 (aq) + H2O (l)
NaHSO4 (aq) + H2O (l)
 Sulfuric acid in Space
Sulfuric acid in SpaceIn 1999, solid (frozen) sulfuric acid was discovered on Jupiter's moon, Europa. Scientists have even identified solid sulfuric acid hydrates such as hemitriskaidecahydrate (H2SO4.6˝H2O).
Aromatic sulfonation is when an H is replaced by SO3H (a sulfonic acid group). In the case of benzene, it needs to be heated with conc. H2SO4 for 8 h to produce benzenesulfonic acid. This reaction is too slow and not to be attempted as benzene is implicated in childhood leukemia.
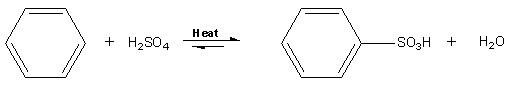
Instead, it's better (faster and safer) to use toluene (methylbenzene), as the methyl (CH3) group is electron releasing and speeds up the reaction. The procedure is: Add 30 drops of conc. H2SO4 to 12 drops of (toluene) in a test-tube. Warm until the methylbenzene has dissolved into the acid layer. Pour the mixture into 30 cm3 of a cold saturated solution of sodium chloride. White crystals of sodium methylbenzene sulfonates are formed. The methyl group is ortho-para directing.

![]()