

|
Business Name |
|
Your business tag line here. |
|
Who receives Herceptin?
· There was much press coverage of Herceptin recently due to the cost of the treatment and therefore who was entitled to it on the NHS.
· The following link concerns a legal case about a woman who won the right to be given Hercetpin: www.irwinmitchell.com
· There have recently been some relevant and interesting journals published concerning herceptin. They can be found on the Web of Knowledge.
· For example Nahta R, Yu DH, Hung MC, et al, Mechanisms of disease: understanding resistance to HER2-targeted therapy in human breast cancer, 2006, 3 (5), p269-280
· History of Herceptin Genentech, the UCLA Medical Center. · Find out more about the research and development behind monoclonal antibodies http://www.antibody.com/index2.html
· Also see, as related article; Booy EP, Johar D, Maddika S, et al. |
|
Who is it for?
Herceptin treatment is only for people with metastatic cancer who have not already had chemotherapy for it. Also where anthracycline treatment is unsuitable.
However Trastuzumab is also licensed to those with metastatic cancer as monotherapy and the patient having had at least two chemotherapy treatments, with the possibility of having had an anthracycline and a taxane. Women with oestrogen receptor positive breast cancer usually should have had HRT.
What are the risks?
As with any treatment or drug there are side effects associated with Herceptin.
The following information can be found in the British National Formulary (47 March 2004).
Cautions
Symptomatic heart failure History of hypertension Coronary artery disease Pregnancy
Contra-Indications
Dyspnoea at rest; breast feeding.
Side Effects
Infusion related side effects, chills, fever, hypersensitivity reactions, for example anaphylaxis urticaria and angioedemea, pulmonary events, cardiotoxicity, gastro-intestinal symptoms, asthenia, headache, chest pains, arthralgia, myalgia, hypotension. |
|
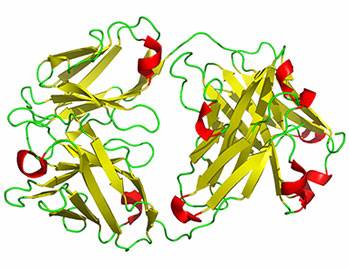
Click on the image to the left to view the molecule in 3D |