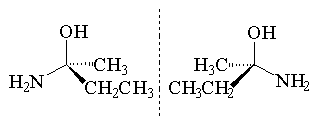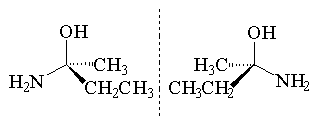
Enantiomers
Enantiomers are a pair of optical isomers. Optical isomerism occurs when a molecule (or ion) and its mirror image are not structurally identical. A molecule that is distinct from its own mirror image, as a left hand is distinct from the right hand, is said to be chiral. A quick way of checking whether an organic compound is chiral is to see whether or not it contains a carbon atom with four different substituents; if this is the case then the molecule is chiral. The enantiomers are labelled either S or R depending on their absolute configuration.
For example the following molecule is enantiomeric since the central atom is bonded to four different substituents thus allowing the existence of two non-superimposable mirror images.