![]()
Ivermectin
The treatment for river blindness
![]()
Simon Cotton
University of Birmingham
Molecule of the Month October 2015
(Updated August 2021)
![]()
IvermectinThe treatment for river blindness
Simon Cotton Molecule of the Month October 2015 (Updated August 2021)
|
 |
 Ivor who?
Ivor who?Not a person, but a molecule.
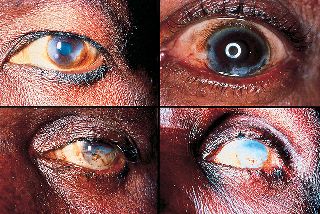
It is a drug for the treatment of Onchocerciasis.
The disease is also known as river blindness. It is sometimes called Robles' disease, after the Guatemalan doctor Rodolfo Robles, who first linked the blindness with an insect a century ago (1915).
The infection is associated with a nematode worm Onchocerca volvulus, which are transmitted by Simulium blackflies which live and breed near fast-flowing streams and rivers. The worms carry parasitic Wolbachia bacteria. The bite of the flies enables the worm larvae to enter the human’s body; after maturing into adults, followed by breeding, the larvae (microfilariae) formed move towards the skin, and release the bacteria when they die. The bacteria trigger an immune response which leads to lesions on the eye and possible blindness (the “river blindness”).

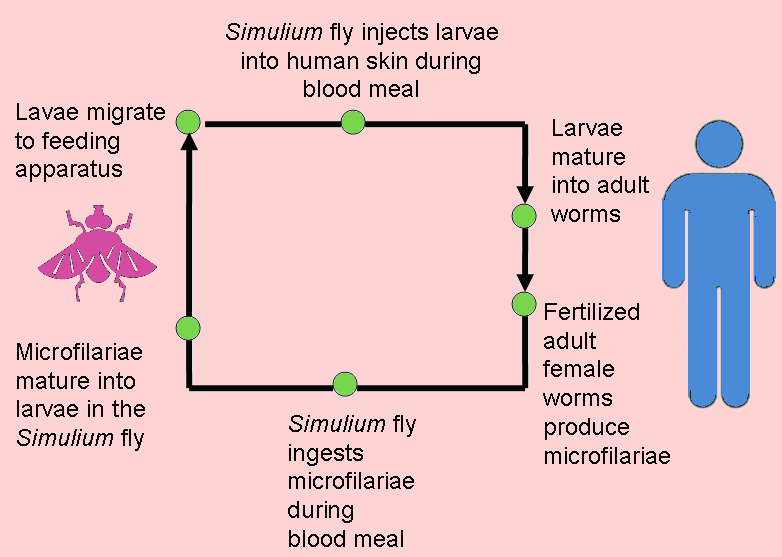
Left: the Simulium fly (from http://flipper.diff.org/app/items/6730).
Right: Simplified life cycle of Onchocerciasis volvulus, modified from the original at: http://emedicine.medscape.com/article/224309-overview#a0104.
 It started with the avermectins. In 1974, a group of researchers headed by Professor Satoshi Ōmura (photo, right) of the Kitasato Institute, isolated an organism with promising antimicrobial properties in a soil sample (sample OS-3153) picked up near a golf course at Kawana, Ito City, Shizuoka Prefecture, Japan. This was passed on to researchers at the Merck, Sharpe and Dohme (MSD) research laboratories in the USA, who isolated a small family of natural products that became known as avermectins. For many years, scientists have looked in soil samples for the source of potential medicines, like the tetracyclines (MOTM September 2011) or streptomycin (MOTM April 2014). There are 8 avermectins, molecules with closely related structures. They are made by fermentation from the bacterium Streptomyces avermitilis.
It started with the avermectins. In 1974, a group of researchers headed by Professor Satoshi Ōmura (photo, right) of the Kitasato Institute, isolated an organism with promising antimicrobial properties in a soil sample (sample OS-3153) picked up near a golf course at Kawana, Ito City, Shizuoka Prefecture, Japan. This was passed on to researchers at the Merck, Sharpe and Dohme (MSD) research laboratories in the USA, who isolated a small family of natural products that became known as avermectins. For many years, scientists have looked in soil samples for the source of potential medicines, like the tetracyclines (MOTM September 2011) or streptomycin (MOTM April 2014). There are 8 avermectins, molecules with closely related structures. They are made by fermentation from the bacterium Streptomyces avermitilis.
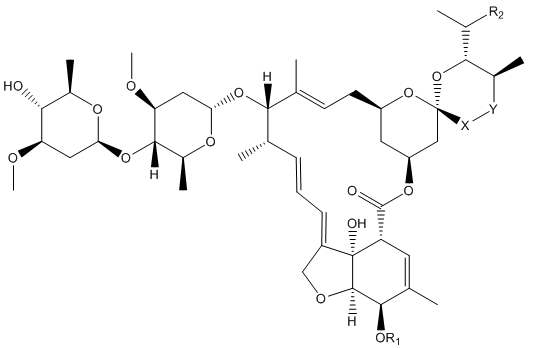
The 8 different avermectins, with the differences between them shown in the table below.
| Name | R1 | R2 | X-Y |
|---|---|---|---|
| Avermectin A1a | Me | Et | CH=CH |
| Avermectin A1b | Me | Me | CH=CH |
| Avermectin A2a | Me | Et | CH2CH(OH) |
| Avermectin A2b | Me | Me | CH2CH(OH) |
| Avermectin B1a | H | Et | CH=CH |
| Avermectin B1b | H | Me | CH=CH |
| Avermectin B2a | H | Et | CH2CH(OH) |
| Avermectin B2b | H | Me | CH2CH(OH) |
The avermectins proved to be have biocidal activity against a wide range of parasites – such as roundworms, lungworms, mites, lice and arachnids; one of these parasites is the tick Rhipicephalus (Boophilus) microplus, one of the most important cattle parasites in tropical regions. Those with the -CH=CH- function are the more active; the most potent was Avermectin B1, occurring as an 80:20 mixture of the similar molecules B1a and B1b, particularly the B1a component. Commercially it is known as Abamectin.
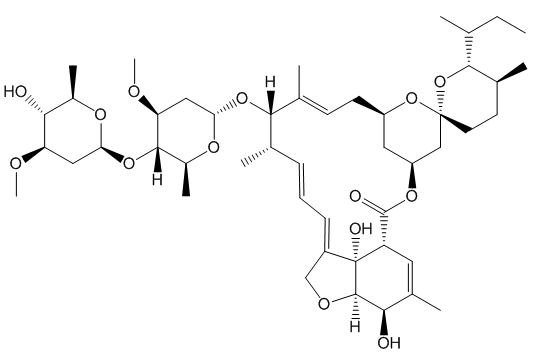
The potency of the avermectins varies, depending on the substituents, so it made sense to look at related molecules. Ivermectin is 22, 23-dihydroavermectin.
You need to add just two hydrogen atoms to reduce a C=C bond in avermectin.
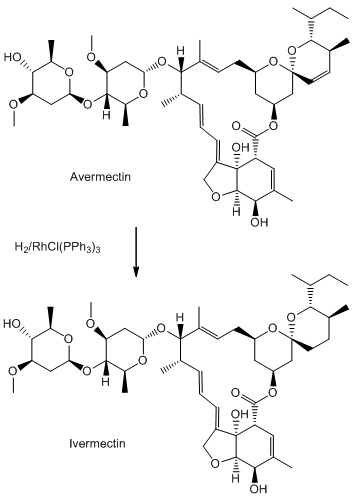
This is done using hydrogen gas, with RhCl(PPh3)3, Wilkinson’s catalyst (MOTM July 2013), to speed things along.
Although there are 5 double bonds in avermectin, three have the E-configuration and only two have the Z- configuration. One of these is much less “hindered” than the other, so it is easier for the catalyst to bind to this one, rather than the others. That is why RhCl(PPh3)3 acts as a selective hydrogenation catalyst.
It’s active against Nemathelminthes or nematodes (roundworms) and the Arthropoda (insects, ticks, and mites). In practice, that has meant it has become a very important veterinary drug to protect cattle, sheep, pigs, horses and dogs from many parasites; for example heartworm infections in dogs.
It was originally thought to work through affecting γ-aminobutyric acid (GABA)-gated Cl- channels but glutamate-gated Cl- channels (which are absent in vertebrates) are now believed to be the main targets; these affect transmission of nerve messages in peripheral muscles of the insects and nematodes. Humans are not affected because ivermectin cannot cross the blood-brain barrier to reach these transmitter sites in the central nervous system; a very few types of dog (e.g. collies) are affected.
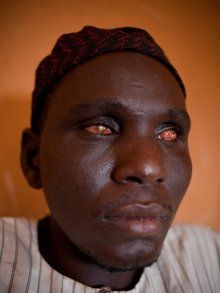 And river blindness?
And river blindness?It is estimated that some 100 million people are at risk of contracting it, mainly in Africa, with some 40,000 new cases of blindness a year, many of these young adults. Ivermectin is believed to kill the microfilariae very gradually, so that the inflammatory response is avoided. It also reduces the chance of the flies being able to transfer them to other potential victims.
Clinical trials on Ivermectin began in 1981 in Senegal. Results were so promising that trials were carried out in several other African countries. In 1987, Merck, the company which had developed Ivermectin, promised that they would donate it – under the brand name Mectizan - free of charge to anyone for as long as it was needed.
The disease has been wiped out in Colombia and there are indications that it is only a matter of time before it is eliminated from Latin America.
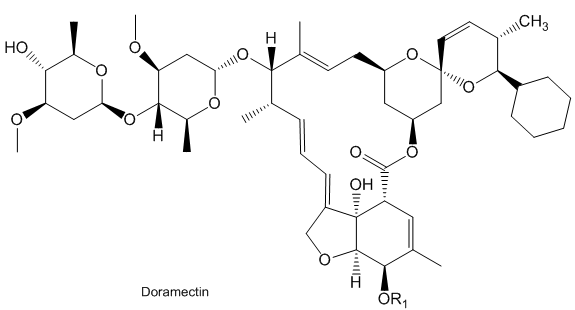
Doramectin is a similar molecule, used to treat parasites in animals, such as cattle, horses, sheep and pigs.
Indeed. It was announced on October 5th 2015 that the 2015 Nobel Prize in medicine was to be shared between William C. Campbell and Satoshi Ōmura “for their discoveries concerning a novel therapy against infections caused by roundworm parasites” – the naturally occurring Avermectin and its development into the semisynthetic Ivermectin – and Youyou Tu “ for her discoveries concerning a novel therapy against Malaria” – Artemisinin (MOTM May 2013).
Ah, you’ve seen the stories in the media! There is no evidence for this claim, but that has not stopped some from advocating it. Ivermectin has been effective against many parasitic infections, but it not been shown to go beyond this. Neither the Food and Drug Administration (FDA) in the USA nor the UK COVID-19 Therapeutics Advisory Panel have permitted its use, and the same situation holds in the European Union. It is possible that scientists could use the structure of ivermectin to suggest how it might attach COVID.
Part of the problem is that scientists have been publishing material on ‘preprint servers’, which means that they are effectively making their claims public before they have been subject to review by their peers, the traditional system. Thus, there have been suggestions of the effectiveness of ivermectin against COVID-19, but scientists have challenged the data reported in a preprint study which claimed that ivermectin was effective in reducing COVID deaths.
A randomised clinical trial has been carried out in Colombia on adults with mild COVID-19, using a 5-day treatment with ivermectin. It was found that, compared with placebo, there was no significant improvement in the time taken to resolve symptoms. The investigators concluded: ‘The findings do not support the use of ivermectin for treatment of mild COVID-19.’
![]()
![]()
![]() Back to Molecule of the Month page. [DOI:10.6084/m9.figshare.5259823]
Back to Molecule of the Month page. [DOI:10.6084/m9.figshare.5259823]