![]()
 |
DESOMORPHINE(a.k.a. Krokodil, the ‘flesh-eating’ drug) |
|
Simon Cotton
Molecule of the Month February 2020
| |
When injected this drug causes horrific skin lesions and ulcers; the skin, muscle and bone near the injection site decay and rot while the drug-user is still alive. As a result it's been called the most dangerous drug in the world.
No, it was first reported in 1932.
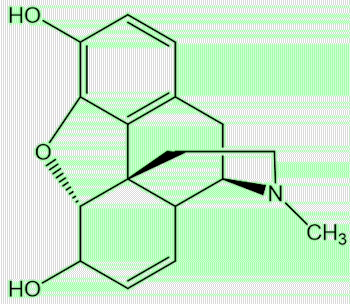
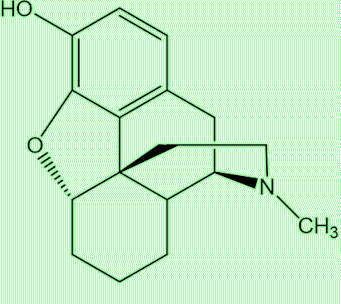
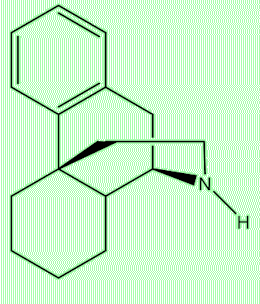
Yes, put the two substances side by side and you can see the similarities. Compounds with this structure based on three fused rings of carbons, one aromatic, with a characteristic nitrogen-containing bridge, are known as morphinans.
 |
 |
 |
| Morphine | Desomorphine (Krokodil) |
Morphinan |
No, it is a ‘semi-synthetic’ substance.
Many natural substances have complicated structures which are hard (and inefficient and expensive) to make from scratch. Often it is possible to use another natural substance with the same core structure as the starting material, in order to make the desired product in just one or two steps. That’s semisynthesis. You are getting Nature to do the hard work of making the ‘difficult’ part of the structure for you, while you just add the finishing touches.
Codeine (MOTM December 2015) is the usual starting material. One way of doing this is to replace the hydroxyl group with a halogen, then use catalytic reduction to remove that and also to reduce the double bond, the one bit of unsaturation outside the benzene ring (whose reduction is of course much more difficult). Last of all, the CH3 is removed from the methoxy group with a reagent such as BBr3.
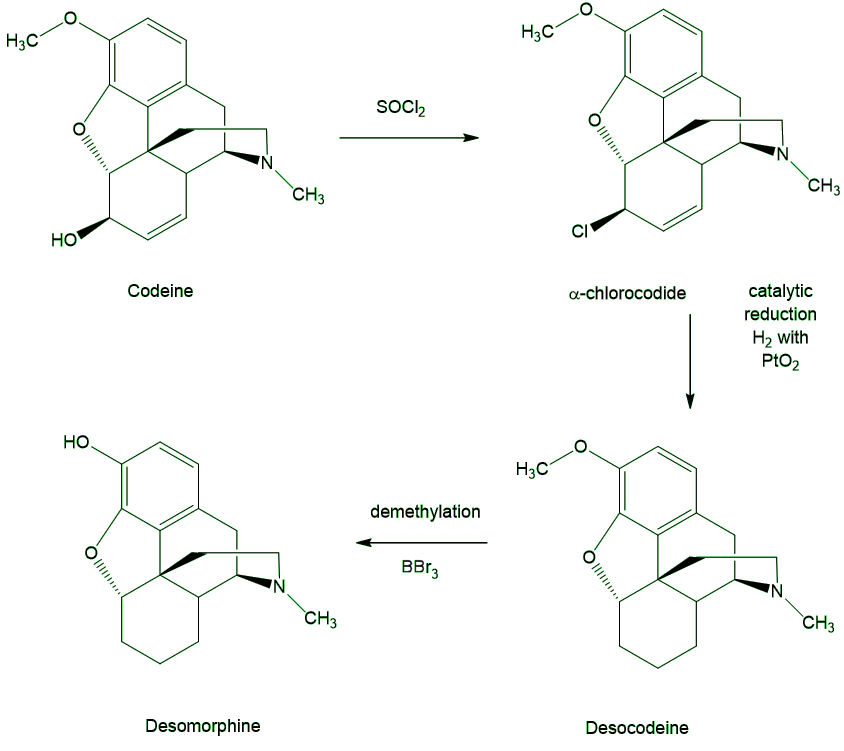
However, addicts use a different process. They first have to extract codeine from the source tablets (or syrup) using an available solvent like petrol, then carry out the reduction, heating the codeine with iodine and red phosphorus in water. This produces HI, which is the actual reducing agent.
Even in those days, painkiller addiction was a problem. Scientists wanted to create a painkiller with fewer side-effects than morphine. Desomorphine was a stronger painkiller than morphine with fewer side effects (e.g. depressing respiration) but it was also found to have a greater potential for dependence, so it was never adopted as a medicine.
Just look back to those structures of desomorphine and morphine (MOTM for November 2004). Desomorphine has one less double-bond and one fewer OH group (morphine has two, desomorphine has one). The more OH (hydroxy) groups a molecule has, the better it will dissolve in water (there are two-OH bonds in a water molecule, remember?). Conversely, the fewer OH groups, the more lipid-soluble a molecule will be, and more likely to cross the blood-brain barrier; thus desomorphine is predicted to access the brain better than morphine. Desomorphine is roughly 10 times stronger a painkiller than morphine; its effects are felt sooner - and it is eliminated from the body faster, leading to a shorter ‘high’, a factor that is important to addicts. In a comparative study with morphine, it was found that its dependence effects lasted only 1/3rd of the time compared with morphine.
But if it wasn’t adopted as a medicine, why are there addicts?Like other countries, Russia has a drug problem. When it started to get harder to obtain heroin on the streets in the early 2000s, someone came up with the idea of using desomorphine. Desomorphine was cheaper than heroin, as it could be made from codeine, which at the time was an easily obtained over-the-counter drug (it ceased to be OTC in 2012), iodine (a common antiseptic) and phosphorus (which could be obtained from matchboxes). It could be prepared in around an hour, even by people with no chemical training, which was important, as addicts needed to inject it every few hours, because of the short-lived ‘high’. But people addicted to other drugs don’t get the symptoms associated with Krokodil!Krokodil means crocodile in Russian, and it got its name because abusers develop scaly skin with a green-black colour, resembling a crocodile. This is caused by the fact that they are injecting (intravenously) a very impure substance, full of toxic and corrosive impurities. The addicts never purify the desomorphine that they make, instead injecting the crude reaction mixture, which contains all sorts of impurities. These cause problems including thrombosis and gangrenous damages, subcutaneous abscesses and ulceration. These result in the flesh rotting, with consequent infection of the bone and muscle, so it has come to be called a “flesh eating” drug. It's also sometimes called the 'zombie drug' because its users resemble zombies with open wounds, ulcers, and skin falling off them in chunks. Another possibility is that morphinans other than desomorphine, which are formed as by-products in the synthesis process, are responsible for some of the nasty side-effects of taking Krokodil, as well as for narcotic effects that cause the victim to ignore them. |
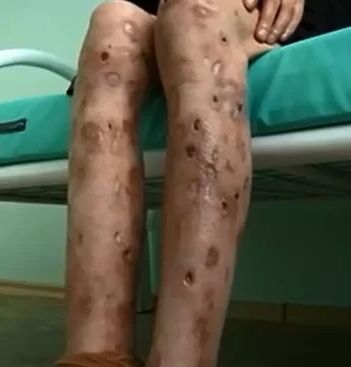 Skin ulcers caused by krokodil use. More graphic images of the sort of horrific damage krokodil use can cause can be seen on medical sites. |
Well, Krokodil addicts are often dead within a year or so, so that is complicating enough. And of course, Krokodil users have the usual risks of sharing syringes whilst being an intravenous injector, namely AIDS/HIV and hepatitis C. But addicts take these risks for the short-lived euphoria they get from an opiate. If they try to come off it, detox takes around a month, compared with the week or so for recovering heroin addicts.
![]()
![]()
![]() Back to Molecule of the Month page. [DOI:10.6084/m9.figshare.11671383]
Back to Molecule of the Month page. [DOI:10.6084/m9.figshare.11671383]