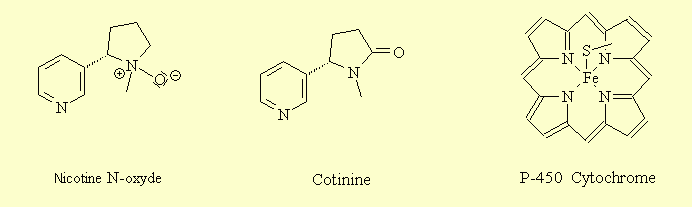
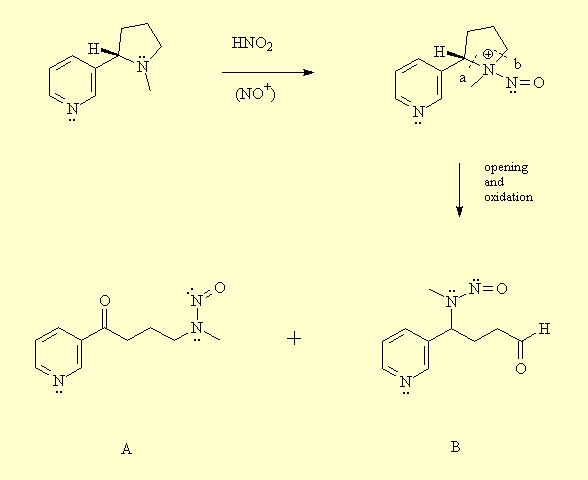
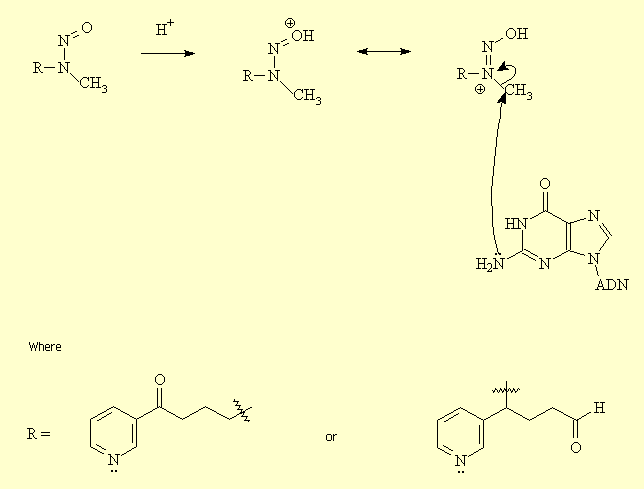
2 - Action on nicotinic receptors
The active form of nicotine is a cation whose charge is located on the nitrogen
of the pyrrole cycle. This active form is very close to acetylcholine.
It has been demonstrated that nicotine interferes with acetylcholine, which
is the major neurotransmitter of the brain. Acetylcholine can bind to two
different kinds of receptors: nicotinic receptors, which are activated
by nicotine, and muscarinic receptors, which are activated by muscarine.
Nicotine and muscarine are thus specific agonists of one kind of cholinergic
receptors (an agonist is a molecule that activates a receptor by reproducing
the effect of the neurotransmitter.)
Nicotine
competitively binds to nicotinic cholinergic receptors. The binding of
the agonist to the nicotinic receptor triggers off a conformation change
of the architecture of the receptor, which opens the ionic channel during
a few milliseconds. This channel is selective for cations (especially sodium).
Its opening thus leads to a brief depolarization. Then, the channel closes
and the receptor transitionally becomes refractory to agonists. This is
the state of desensitization. Then, the receptor usually goes
back to a state of rest, which means that it is closed and sensitive to
the agonists. In case of continuous exposure to agonists (even in small
doses), this state of desensitization will last long (long-term inactivation).
Operating cycle of a nicotinic receptor:
Physiological normal conditions: After the opening of the canal by
binding to acethylcholine, the receptor becomes desensitized before it
goes back to the state of rest or it is regenerated. Continuous exposure to tobacco: Nicotine substitutes for acetylcholine
and over stimulates the nicotinic receptor. Then, the receptor is long-term
inactivated and its regeneration is prevented by nicotine.
Nicotine activates dopamine systems within the brain.
Dopamine is a neurotransmitter which is directly responsible for mediating
the pleasure response. Nicotine triggers off the production of dopamine
in the nucleus accumbens. A prolonged exposure
of these receptors to nicotine reduces the efficiency of dopamine by cutting
down the number of available receptors. Consequently, more and more nicotine
is needed to give the same pleasurable effect.
After
a brief period of abstinence (overnight for instance), the brain concentration
of nicotine lowers and allows a part of the receptors to recover their sensibility.
The return to an active state rises the neurotransmission to an abnormal
rate. The smoker feels uncomfortable, which induces him to smoke again. The
first cigarette of the day is the most pleasant because the sensibility
of the dopamine receptors is maximal. Then, the receptors are soon
desensitized
and the pleasure wears off. This is
the vicious circle
of smoking.
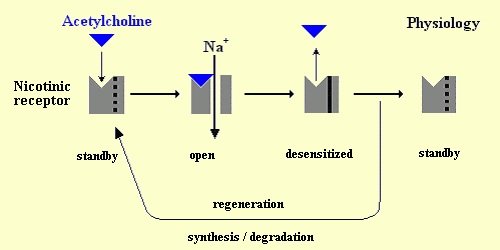
[Figure reproduced courtesy of Dr. Sylvain Bartolami, INSERM U1198 "Mécanismes Moléculaires dans les Démences Neurodégénératives", Eq. 5 "Integrative Biology of Neuroregeneration", Université de Montpellier, France].
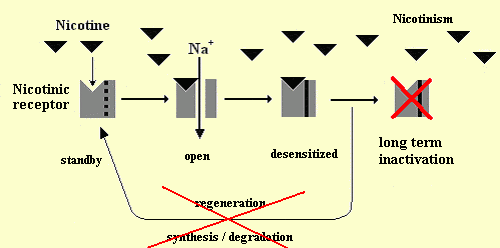
[Figure reproduced courtesy of Dr. Sylvain Bartolami, INSERM U1198 "Mécanismes Moléculaires dans les Démences Neurodégénératives", Eq. 5 "Integrative Biology of Neuroregeneration", Université de Montpellier, France].
3 - Tolerance and dependence
on nicotine