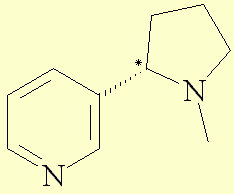
Nicotine is also called 3-(1-methyl-2-pyrrolidinyl)pyridine according to the IUPACa nomenclature. It is a bicyclic compound with a pyridine cycle and a pyrrolidine cycle. The molecule possesses an asymetric carbon and so exists in two enantiomeric compounds. In nature, nicotine only exists in the S shape, which is levogyre.

Representationb of the R molecule in 3D :

| Formula | C10H14N2 |
| Molecular Weight | 162,234 g.mol-1 |
| Melting point | -79 ° C |
| Boiling point | 247 ° C |
| Rotatory index (S) |
aD
= -168 at 20° C |
| Density | d=1,010 |
| Refractive index | n=1,530 |
| Comments |
Pale yellow to dark brown liquid with a slight, fishy odor when warm.
Insecticide. |
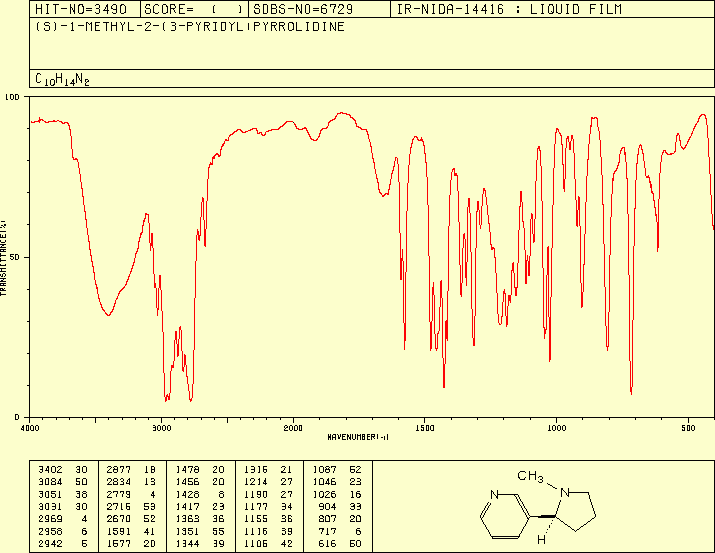
On this
spectrum, we can notice several peaks, which characterise the different
chemical functions of nicotine:
· Around 3400 cm-1, we can see the large peak
of water (it deals with a liquid film).
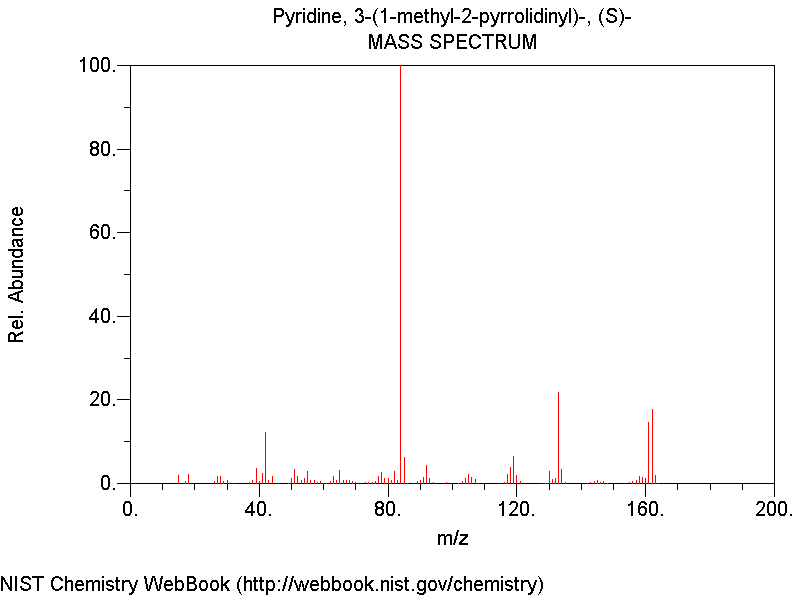
We can notice the molecular peak at 162 m/z. However, the biggest peak is at 84 m/z. It corresponds to the fragmentation of nicotine. It deals with the pyrrolidine cycle, which has this molecular weight. During the electronic bombardment, the nicotine was split between the two cycles.
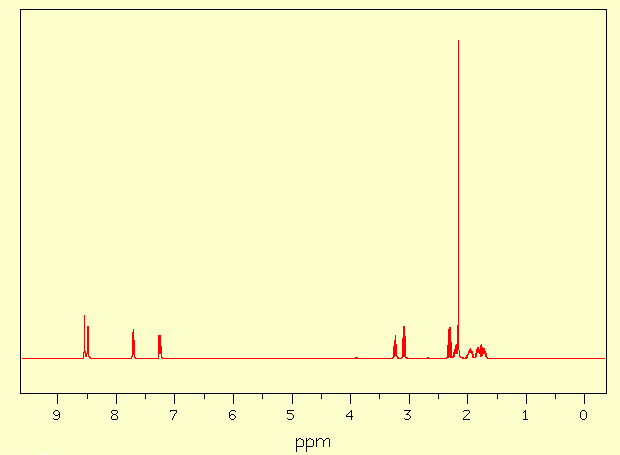
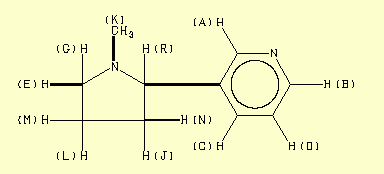
Assignment Chemical shift (ppm) A 8,543 B 8,487 C 7,711 D 7,255 E 3,237 F 3,087 G 2,307 J 2,200 K 2,160 L 1,958 M 1,820 N 1,725 Done with C-H
COSY
148,51
· Between 2970 and 2780 cm-1 : C-H stretching.
· The peak at 1677 cm-1 : aromatic C=N double bond stretching.
· The peak at 1691 cm-1 : aromatic C=C double
bond stretching.
· The peaks at 717 cm-1 and 904 cm-1 correspond to the out of
plane bending of the C-H bond of the monosubstituted pyridinic cycle.
4 - Mass spectrum analysis
5 - 1H-RMN analysis in CDCl3 (400MHz)
6 - 13C-RMN analysis in CDCl3
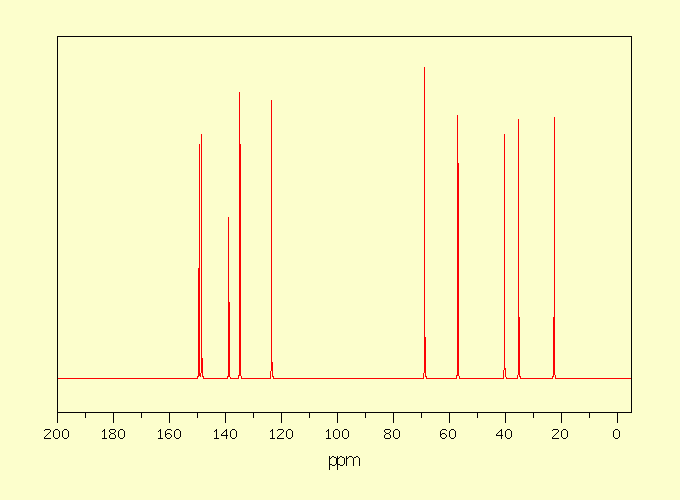
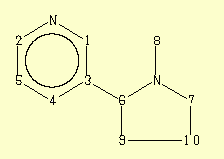
Assignment
Chemical
shift (ppm)
Integration
1
149,45
750
2
783
3
138,80
517
4
134,86
917
5
123,54
894
6
68,83
1000
7
56,96
844
8
40,32
783
9
35,23
833
10
22,60
839
The IR and RMN spectra were found on the site of SDBSc