![]()
Psilocybin
(and Mescaline)
The hallucinogens in 'magic mushrooms'
![]()
![]()
Molecule of the Month October 1999
(Updated July 2017)
Also available: JSMol version.
![]()

|
Psilocybin(and Mescaline)The hallucinogens in 'magic mushrooms'
Molecule of the Month October 1999
|
 |
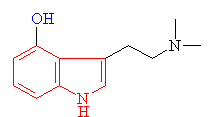
Psilocybin is the main active ingredient in so-called 'magic mushrooms'. It has hallucinogenic properties, and is closely related to mescaline in structure. Both chemicals have been known for centuries by the Aztecs in Mexico, who used them in tribal rites, believing the vivid, colourful hallucinations had religious significance. Indeed, the mushroom was so important to the Aztecs that they named it teonanácatl, meaning "God's flesh" or "God's meat". This mushroom was said to have been distributed to the guests at the coronation of Montezuma to make the ceremony seem even more spectacular. The Aztecs even had professional mystics and prophets who achieved their inspiration by eating other hallucinogenic plants, such as the mescaline-containing peyote cactus (Lophophora williamsii). During the 19th Century, the use of peyote cacti in tribal rituals spread north to the natives of North America, such as the Comanches, Kiowas and the Mescalero Apaches, from where mescaline obtained its name. Over the years, the use of these drugs in religious rites became fused with Christianity, and even today some tribes believe that God put some of His power into peyote, and Jesus was the man who gave the plant to the Indians in a time of need.
 |
 |
| Psilocybe semilanceata (Fries) Kummer - one of the family of 'magic mushrooms'. |
The peyote cactus. Mescaline is made when the caps, or buttons, are dried and eaten, brewed into tea or powdered into capsules. |
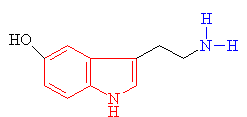
Psilocybin itself, comes from 'magic' mushrooms (such as Psilocybe mexicana and Stropharia cubensis), several varieties of which grow in temperate regions of the world, including the USA, Europe and the UK. Their use became fashionable in the UK in the 1970s as a 'natural' and legal alternative to LSD. In most countries it is not illegal to possess or consume these mushrooms, however if they are prepared (e.g. crushed or dried), or deliberately cultivated, they then become a Class A drug punishable by imprisonment.
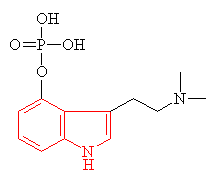 |
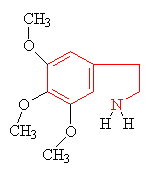 |
| Psilocybin The indole ring is shown in red. |
Mescaline The structure resembling an indole ring is shown in red. |
Psilocybin and mescaline are psycho-active because they closely resemble the structures of neuro-transmitters that convey impulses from one nerve to another, especially in the brain. Examples are serotonin and norepinephrine. The hallucinogenic molecules fit into the same receptors as the neuro-transmitter, and over-stimulate them, leading to false signals being created.
 |
 |
| Serotonin The indole ring is shown in red and the amine group in blue. |
Psilocin The hydrolysed form of psilocybin with the change shown in blue. |
Many hallucinogens are variations of important biological substances called indole-amines. These contain an indole ring structure (highlighted in red in all the diagrams), which is simply a 6-membered benzene ring fused to a 5-membered ring containing nitrogen. An example of an important indole-amine is the serotonin molecule mentioned earlier. The main variation is to add a few methyl groups, making the molecule more lipophilic (soluble in fats), and so better able to penetrate the fatty membranes that protect nerves and nerve endings. This allows the molecules to more readily penetrate the central nervous system, and hence makes them more potent. Psilocybin has this indole ring, with a phosphoric acid group attached to it. In the body the phosphoric acid group is oxidised to the hydroxyl compound, known as psilocin, which is equally psycho-active. Mescaline does not have the indole ring, but as shown in red in the diagram above, its structure can be represented so as to suggest its relation to the ring. The most famous hallucinogenic drug, LSD has a complicated structure, but it, too, is based around the indole ring.
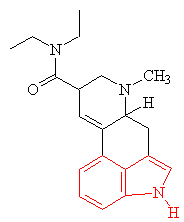 |
| Lysergic acid diethylamide - LSD The indole ring is highlighted in red. |
Do these drugs produce “religious experiences”?Aldous Huxley certainly seemed to think so. In his book ‘Heaven and Hell’ he says, “Reading these accounts [of drug-induced visionary experiences], we are immediately struck by the close similarity between induced or spontaneous visionary experience and the heavens and fairylands of folklore and religion”. Recent double-blind studies support the view that psilocybin can induce experiences similar to mystical experiences and enhance autobiographical recollection. It has potential in psychotherapy either to facilitate the recall of salient memories or to reverse negative cognitive biases. There is evidence that a strong dose of psilocybin can affect personality for a year, possibly for longer. And if psilocybin can produce religious experiences, then it’s quite possible that naturally occurring psychoactive molecules like dopamine or serotonin could do the same, if their concentrations in the brain become unusually low or high for some reason. It could be argued that many of the ancient prophets and mystics revered by different religious all over the world just experienced an imbalance in their brain chemistry, rather than a true supernatural intervention. Either way, psilocybin and other psychoactive molecules are definitely food for thought (literally, one might say!). |
 The Bernini's statue of the Ecstasy of St Teresa in Rome. This statue is meant to represent the moment of religious ecstasy when encountering an angel. Or was it an hallucination due to an imbalance in brain chemistry? |
![]()
![]()
![]() Back to Molecule of the Month page. [DOI:10.6084/m9.figshare.5245549]
Back to Molecule of the Month page. [DOI:10.6084/m9.figshare.5245549]