
![]()
THE MIGHTY QUINiNe
![]()
Simon Cotton
Uppingham School, Rutland, UK
![]()
Molecule of the Month July 2005
Also available: JSMol version.
![]()
 |
THE MIGHTY QUINiNe
Simon Cotton
Molecule of the Month July 2005
|
 |
Well, quinine is a mighty molecule, and once again, it's an important drug.
For many years quinine was the drug of choice in treating malaria. Malaria is reckoned to have killed more people in World War II than either bullets or bombs. Even today, it causes over a million deaths a year.
 A female Anopheles gambiae mosquito feeding on a person. Anopheles transmit the malaria parasite. Photo courtesy of CDC, viewed at National Institute of Allergy and Infectious Diseases. |
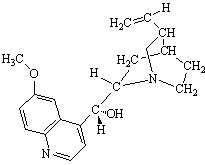 Quinine |
 How?
How?Malaria is a disease caused by protozoa, transmitted by the female Anopheles mosquito. It was once thought to be due to "bad air", hence the name malaria derived from an Italian phrase (mal aria).
 How long has quinine been known?
How long has quinine been known?16th century Spanish settlers in South America found natives had used the powdered bark (right) from the Cinchona tree (left) to treat fevers for hundreds of years; Jesuits brought the bark back to Europe around 1630. Quinine was isolated from the bark in 1820.
 Patchily. Malaria was widespread in Northern Europe from the 15th to the 19th centuries. Known as the "Ague", it was endemic in parts of England like the Cambridgeshire Fens and the Kent and Essex marshes. Its disappearance is probably due to factors like improved marsh drainage and better housing and sanitation. It is said that Oliver Cromwell's (right) final illness in 1658 was fatal as he refused to take "Jesuit's bark" because of Protestant suspicion of something associated with Roman Catholics. Demand for quinine grew with African and Asian colonialisation; cuttings taken from Peru led to a large quinine-based industry in Java. Quinine was used worldwide. Undertakings like building the Panama Canal could not have happened without it.
Patchily. Malaria was widespread in Northern Europe from the 15th to the 19th centuries. Known as the "Ague", it was endemic in parts of England like the Cambridgeshire Fens and the Kent and Essex marshes. Its disappearance is probably due to factors like improved marsh drainage and better housing and sanitation. It is said that Oliver Cromwell's (right) final illness in 1658 was fatal as he refused to take "Jesuit's bark" because of Protestant suspicion of something associated with Roman Catholics. Demand for quinine grew with African and Asian colonialisation; cuttings taken from Peru led to a large quinine-based industry in Java. Quinine was used worldwide. Undertakings like building the Panama Canal could not have happened without it.
 Gin was invented by a Dutchman in the mid-17th century; he flavoured spirits with oil of juniper (French geničvre) calling it genever, which in time was corrupted to gin. Quinine is almost insoluble in water (about 0.05 g per 100 ml water) but it is readily soluble in ethanol. It is alleged that the British Empire-builders took their daily dose of quinine dissolved in ethanol in the form of gin, using lemon or lime to help mask the bitter taste of the quinine. Tonic water came on the scene later (Schweppes introduced it around 1870). There is about 20 mg of quinine per 6 fluid ounces of one leading brand.
Gin was invented by a Dutchman in the mid-17th century; he flavoured spirits with oil of juniper (French geničvre) calling it genever, which in time was corrupted to gin. Quinine is almost insoluble in water (about 0.05 g per 100 ml water) but it is readily soluble in ethanol. It is alleged that the British Empire-builders took their daily dose of quinine dissolved in ethanol in the form of gin, using lemon or lime to help mask the bitter taste of the quinine. Tonic water came on the scene later (Schweppes introduced it around 1870). There is about 20 mg of quinine per 6 fluid ounces of one leading brand.
The recommended daily dose of quinine for malaria is 600 mg, three times daily, for up to a week. At that rate you would be drinking up to 100 gin and tonics a day. This would certainly cure you of malaria, but could cure you of life first. Nice try, though.
The flat ring fits between the bases in DNA and interferes with transcription, blocking cell replication.
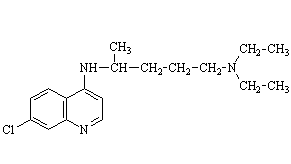
Drugs like mepacrine and chloroquine were developed to counter quinine shortages. When the Japanese invaded Java in 1941, quinine supplies to most of the world stopped, so these alternatives were welcome.
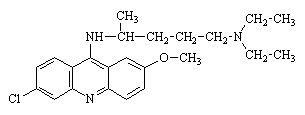 |
 |
| Mepacrine | Chloroquine |

 They had disadvantages. Mepacrine turned the skin yellow. "Tokyo Rose", an American-born and educated radio broadcaster (her real name was Ikuko Toguri; she was a UCLA graduate, photo left) who broadcast Japanese propaganda to American troops, told them that mepacrine would also make them sterile. The result was that many American troops did not take their antimalarial tablets, and caught malaria. A long and expensive campaign featuring "Annie the mosquito" (photo, right) was required to persuade the soldiers to take antimalarial precautions. Chloroquine was better, but as time went on, the malarial parasite developed a resistance to chloroquine.
They had disadvantages. Mepacrine turned the skin yellow. "Tokyo Rose", an American-born and educated radio broadcaster (her real name was Ikuko Toguri; she was a UCLA graduate, photo left) who broadcast Japanese propaganda to American troops, told them that mepacrine would also make them sterile. The result was that many American troops did not take their antimalarial tablets, and caught malaria. A long and expensive campaign featuring "Annie the mosquito" (photo, right) was required to persuade the soldiers to take antimalarial precautions. Chloroquine was better, but as time went on, the malarial parasite developed a resistance to chloroquine.
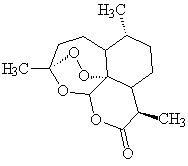
Yes it is still used to treat severe falciparum malaria. The big development has been Chinese drugs; herb extracts (qinghao) have long been used to treat fevers; an extract called Artemisinin (qinghaosu) works against chloroquine-resistant malaria. Molecules developed from it, like arte-ether, seem even better.
These molecules contain a peroxide (O-O) group. In the presence of iron from damaged blood cells, the peroxide group is believed to generate reactive free radicals These may destroy the DNA of the plasmodium.
 |
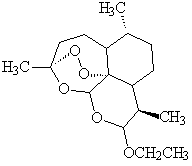 |
| Artemisinin | Arte ether |
One recent development has been a substance known as Fosmidomycin. It blocks an enzyme called 1-deoxy-D-xylulose-5-phosphate (DOXP) reductoisomerase. In the malarial parasite, this is involved in making molecules in the isoprenoid family. Humans make isoprenoids by a different pathway, so Fosmidomycin is toxic to the malarial parasite but not to people. It is possible that Fosmidomycin might be used in combination with another drug to help it work quicker and also to lessen the chance of drug resistance developing.
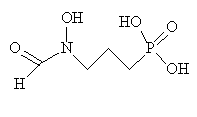 |
| Fosmidomycin |
Another drug in the news as a possible antimalarial is OZ277. It has been developed by an international team of scientists, working at the University of Nebraska, at Monash University in Australia, the laboratories of Hofman La Roche in Basel, Switzerland and at the Swiss Tropical Institute.
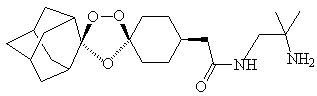 |
| OZ277 |
It is an attempt to improve on Artemisinin.
Because it is a plant extract, Artemisinin is expensive, and is also quickly broken down in the body, so frequent doses are needed. OZ277 contains the type of bridge which is believed important to the activity of Artemisinin (it generates radicals that target proteins in the parasite), and was engineered with the bulky adamantane group next to it to protect the peroxide bridge. Other requirements of the drug are low toxicity and the need for only one daily dose, by mouth, and effectiveness within a three-day course of treatment, as well as good water solubility. OZ277 is more active than Artemisinin; it has sufficient solubility to be administered orally, and looks cheap to produce. It has a longer lifetime in the plasma, so it stays active longer in the body. It is currently in Phase I clinical trials in the UK, and is due to be tried on malaria patients in 2005. It will be used in combination with some other drug in order to reduce the likelihood of drug resistance.
Despite global warming on the agenda and fears of malaria spreading north, Quinn will still probably just need quinine for his gin and tonic.
![]()
 Malaria
Malaria![]()
![]() Back to Molecule of the Month page. [DOI:10.6084/m9.figshare.5245840]
Back to Molecule of the Month page. [DOI:10.6084/m9.figshare.5245840]