![]()
SKATOLE
The smell of human excrement
![]()
Simon Cotton
Uppingham School, Rutland, UK
![]()
Molecule of the Month April 2006
Also available: JSMol version.
![]()

 |
SKATOLEThe smell of human excrement
Simon Cotton
Molecule of the Month April 2006
|
 |
The smell of human excrement.
Actually, skatole is an important molecule, and crops up in some surprising places.
Ice cream, would you believe?
It is used in very small amounts as a flavouring material in ice cream. The skatole put in ice-cream is man-made. No, let me rephrase that. The skatole put in ice-cream is synthetic, it has no human connections.
 How can it be used in ice-cream?
How can it be used in ice-cream?The odour of skatole depends on its concentration. At low concentrations, skatole actually has a rather nice, sweet smell; it crops up in orange blossom and jasmine in small amounts. It's also used by Zantedeschia aethiopica.
It's an Arum lily, (photo, right), which makes skatole attractive to pollinating insects. It has flowers on the central part (the spadix) in male and female zones. The faint scent attracts crawling insects and bees which pollinate the flowers.
It's used in perfume too.
It is used as a fixative in perfumes.
Perfumes are mixtures of molecules, generally with Molecular Mass below 300, of varying volatilities. Their "top note" is made of volatile substances with instant impact; once this disappears, the "middle note" is longer lived, whilst the "base note" which may linger for days or weeks, and may make up 50% of the perfume, are deep odours that may not be smelt for some time. The fixative is a molecule with a deep odour that has a low volatility and which helps to reduce evaporation of the more volatile components (like musk). This means that the perfume keeps longer.
 How did that use come about?
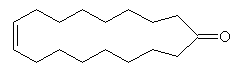
How did that use come about?Civet cats (photo, right), mainly found in Africa and India, produce an oily substance called "civet" from the perineal glands in their abdomen; it is used to mark their territory. Civet has a strong smell, due partly to skatole, and also to civetone (9(Z)-cycloheptadecenone). When diluted, its smell becomes more musky (and less objectionable); demand from the perfume industry for civet has been met by a synthetic version. Chinese civet cats attracted publicity recently as they were (incorrectly) linked with SARS outbreaks.
 |
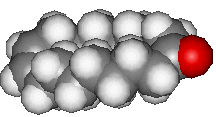 |
| Civetone | ...and its spacefill representation |
It is supposed to be more unpleasant than mercaptans, with a threshold for detection of 1.7x107 molecules per cm3. This sounds a lot, but it is over two orders of magnitude less than pyridine, which itself is pretty objectionable.
 What does skatole look like?
What does skatole look like?Pure skatole is a white crystalline material, believe it or not.
It is derived from the Greek σκωτ or σκατ (skat), meaning dung. It gives meaning to the adjective scatological, meaning filthy or obscene.
Skatole produced by male pigs can cause "boar taint", which makes pork from these sources unattractive as food.
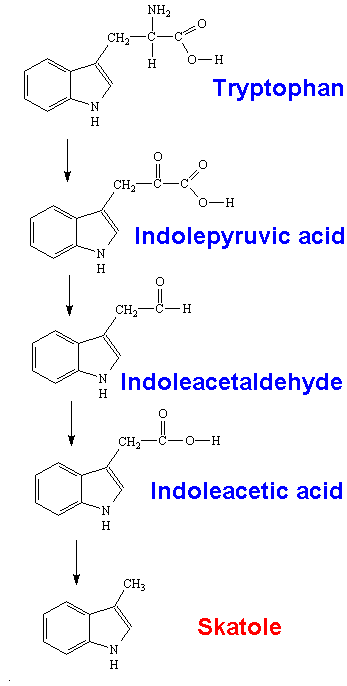
It is produced in the digestive tract by bacterial breakdown of the amino acid tryptophan.
No.
![]()
![]()
![]() Back to Molecule of the Month page. [DOI:10.6084/m9.figshare.5248984]
Back to Molecule of the Month page. [DOI:10.6084/m9.figshare.5248984]