School of Chemistry
University of Bristol
 |
 |
| By Neil Edwards
School of Chemistry |
![]()
"Taxol" is used in these pages to refer to the drug that now has the generic name "paclitaxel", and the registered tradename "Taxol ®" (Bristol-Myers Squibb Company, New York. N.Y.)
JSMol version also available.
![]()
Taxol
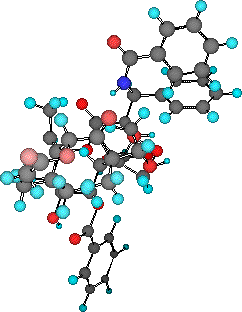
In the early 1960s, the National Cancer Institute (NCI) in the United States initiated a programme of biological screening of extracts taken from a wide variety of natural sources. One of these extracts was found to exhibit marked antitumour activity against a broad range of rodent tumours. Although this discovery was made in 1962, it was not until five years later that two researchers, Wall and Wani, of the Research Triangle Institute, North Carolina, isolated the active compound, from the bark of the Pacific yew tree (Taxus brevifolia). In 1971, Wall a nd Wani published the structure of this promising new anti-cancer lead compound, a complex poly-oxygenated diterpene (shown below).
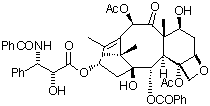
| Taxol - the 3D structure in pdb format can be viewed (using a suitable helper application such as Rasmol) by clicking on the image. |
Biological Profile
Despite its well documented biological activity, very little interest was shown in taxol until scientists at the Albert Einstein Medical College reported that its mode of action was totally unique. Until this finding in 1980, it was believed that the cytotoxic properties of taxol were due to its ability to destabilise microtubules, which are important structures involved in cell division (mitosis). In fact, taxol was found to induce the assembly of tubulin into microtubules, and more importantly, that the drug actually stabilises them to the extent that mitosis is disrupted. Such a novel mode of action was be lieved to make taxol a prototype for a new class of anticancer drugs.
 |
Renewed interest in taxol led to major problems, since many groups wished to conduct clinical trials, and so large quantities of this material were required. The natural source, the Pacific yew tree (left), is an environmentally protected species, which is also one of the slowest growing trees in the world. Isolation of the compound, which is contained in the bark, involves killing the tree, and the quantities available by this method are pitifully small. It would take six 100-year old trees to provide enough taxol to treat just one patient. |
Luckily, a closely related analogue of taxol, called baccatin III was discovered in the leaves of a European species of ornamental shrub Taxus baccata. Although the extraction and subsequent chemical elaboration of baccatin III to taxol was very laborious, the source was renewable, and sufficient quantities were obtained to carry out clinical trials. Results, published in 1989 showed that taxol exhibits very promising activity against advanced ovarian cancer, and in 1992, the US Food and Drug Administration (FDA) approved taxol for the treatment of this condition. The treatment of other types of cancer also looks promising, and in 1994, taxol was approved for breast cancer.
The relatively non-toxic properties of taxol have made it a leading light in the treatment of cancer in the 1990s, providing a non-intrusive alternative to the more radical techniques of radiotherapy and surgery. The cost of producing sufficient quantities of this new wonderdrug, however, is a severely limiting factor. Synthetic organic chemistry may provide a solution to this problem in the years to come.
Total Synthesis of Taxol
Taxol represents an enormous challenge to the ingenuity and creativity of the synthetic organic chemist.
Three total syntheses have been carried out to date. The Holton group and Nicolaou group published their approaches in 1994, and very recently, Danishefsky and co-workers reported their route to taxol. Many other groups worldwide are continuing to carry out research in this area, in order to develop newer and shorter routes to this natural product, but also with a view to creating modified structures which may be more biol ogically active. Such groups include the Magnus researchers at Austin, Texas, and the Wender group at Stanford who have published their results recently, amongst others.
Holton used (-)-borneol as his starting material, which he converted to an unsaturated ketone over 13 chemical steps. This ketone was converted into β-patchouline oxide
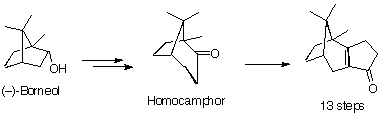
which was epoxidised and treated with a Lewis acid, which induced a skeletal rearrangement, providing tertiary alcohol. This alcohol was again epoxidised, and underwent a fragmentation reaction to create the A and B rings of taxol. The C- ring was introduced using the Robinson-Stork annulation methodology.
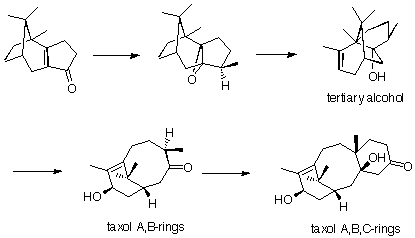
These reactions outline the general route that Holton employed to synthesise taxol. The full synthesis can be found in J.Am.Chem.Soc. (1994) 116, 1597 and 1599.
The route utilised by Nicolaou was a convergent (rather than a linear) route, the A- and C-rings being constructed separately, and then linked together using a Shapiro reaction to connect the southern part, and a McMurray coupling reaction to complete the B-ring.
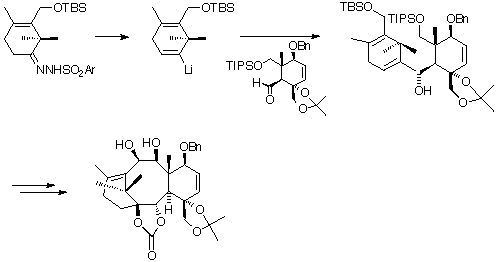
Having created the fused A, B and C ring system, Nicolaou then went on to complete the total synthesis of taxol. For more detailed information see Nature (1994) 367, 630; J.Chem.Soc. Chem.Commun. (1994) 295 and J.Am.Chem.Soc. (1994) 116, 1591.
The method employed by Danishefsky started with the Wieland-Miescher ketone, which was elaborated to a complex enol triflate, bearing an olefin on the C-ring for development to taxol via an intramolecular Heck reaction.
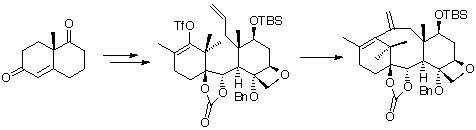
Further details about Danishefsky's route to taxol can be found in J.Am.Chem.Soc. (1996) 118, 2843.
Other Taxol Links
Here are some other good sources of information about taxol:
These are just a selection of the many taxol-related links on the World Wide Web. To find out more, try searching for taxol using any of the available search engines.
![]()
 |
Last updated 7.12.96 |
![]() Back to Molecule of the Month page.
Back to Molecule of the Month page.