
Geometric Isomerism
Geometric Isomerism results most commonly from Carbon-Carbon double bonds. The
important property which introduces the feature is the inability of the Carbon atoms to rotate relative to one another about the
double bond. This is due specifically to the Pi bond but I won't discuss this part of the subject further on this site.
The lack of rotation means the same groups can be attached in different ways to achieve diastereomers. The molecules have identical
connectivity so can't be described as structural isomers.


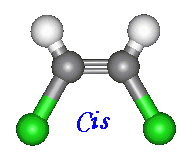
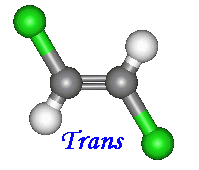
There are two ways to name compounds of this kind. When two of the four groups attached to the carbons
involved are hydrogen, the cis and trans notation can be used. When the other two constituents are on the same side
of the molecules name is given the prefix 'cis'. Otherwise if the constituents are on opposite sides the prefix is 'trans'.
What If You Don't Have Two Hydrogen Constituents?
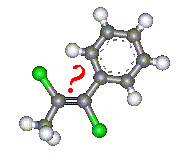 With molecules such as that shown in the image, it is not possible to assign a cis or trans prefix which
unambiguously describes the molecule. Therefore there is a second a second convention which can be used to name any geometric
isomer. This gives us the E and Z isomers.
With molecules such as that shown in the image, it is not possible to assign a cis or trans prefix which
unambiguously describes the molecule. Therefore there is a second a second convention which can be used to name any geometric
isomer. This gives us the E and Z isomers.
Each substituent of the double bond can be given a priority. By looking at the highest priority groups on each of the carbons
we say if both are on the same side (as with cis) this is the 'Z' isomer. If they are on opposite sides we say it is the 'E' isomer
.
Priority is determined by the atomic number of the atoms in the groups. Atoms of higher atomic number are given higher priority. If the
first atoms are the same, you move to the next atom, and keep doing so until a difference can be found.
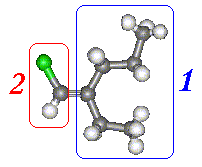 On Carbon 1 we have Ethyl and a Propyl groups. Moving away from the Carbon in the double bond the first difference is at the
second carbon atom. The group at the top has a Carbon, whereas the group at the bottom has a hydrogen. The Carbon is clearly
of higher atomic number and, therefore, priority.
On Carbon 1 we have Ethyl and a Propyl groups. Moving away from the Carbon in the double bond the first difference is at the
second carbon atom. The group at the top has a Carbon, whereas the group at the bottom has a hydrogen. The Carbon is clearly
of higher atomic number and, therefore, priority.
On Carbon 2 there is a Chlorine on the top and a Hydrogen at the bottom. The Chlorine is the higher priority group.
As both high priority groups are on the same side this is the 'Z' isomer.
There are also examples of this sort of isomerism where no double bond is present. One example of this is
disubstituted cycloalkanes. There is no double bond but the rigidity introduced by the ring introduces geometric isomerism.
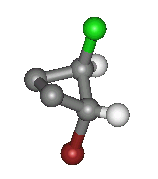
Here the Bromine is pointing down from the ring and the Chlorine is pointing up. This is another type of geometric isomerism,
the molecule shown is the 'trans' form. Hydrogens have been left off this image to make it clearer to see the sites of interest.
 On Carbon 1 we have Ethyl and a Propyl groups. Moving away from the Carbon in the double bond the first difference is at the
second carbon atom. The group at the top has a Carbon, whereas the group at the bottom has a hydrogen. The Carbon is clearly
of higher atomic number and, therefore, priority.
On Carbon 1 we have Ethyl and a Propyl groups. Moving away from the Carbon in the double bond the first difference is at the
second carbon atom. The group at the top has a Carbon, whereas the group at the bottom has a hydrogen. The Carbon is clearly
of higher atomic number and, therefore, priority.


 With molecules such as that shown in the image, it is not possible to assign a cis or trans prefix which
unambiguously describes the molecule. Therefore there is a second a second convention which can be used to name any geometric
isomer. This gives us the E and Z isomers.
With molecules such as that shown in the image, it is not possible to assign a cis or trans prefix which
unambiguously describes the molecule. Therefore there is a second a second convention which can be used to name any geometric
isomer. This gives us the E and Z isomers.