 |

TRICLOSAN
The antibacterial molecule found in soaps, detergents,
toys and surgical cleaning treatments that may
be becoming an enviromental problem

Simon Cotton
University of Birmingham

Molecule of the Month November 2014
Also available: JSMol version.

|
I’ve heard of Triclosan, it’s an environmental pollutant, isn’t it?
Why do you say that?
I’ve seen pictures like this one.
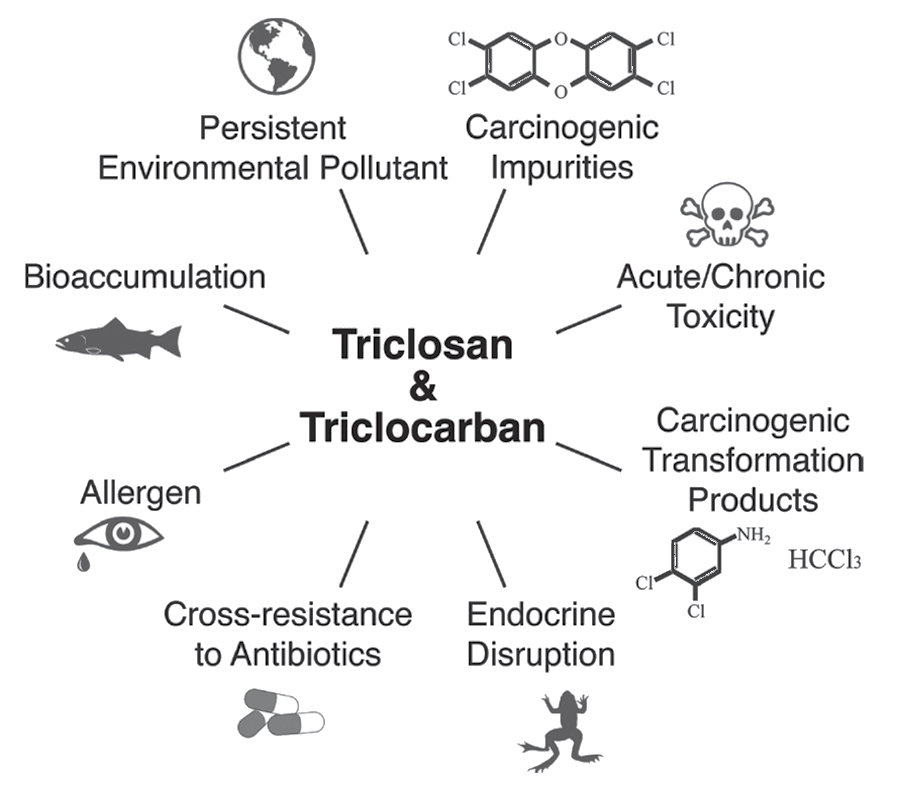
Potential dangers of triclosan [originally from dx.doi.org/10.1021/es500495p]
I see what you mean, but there is more to Triclosan than that.
So what does triclosan do?
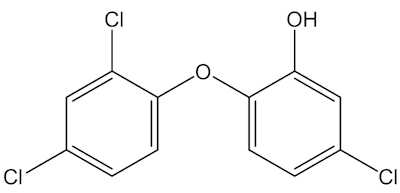
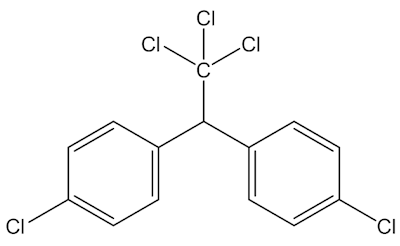
It was originally made by Ciba, and introduced as an insecticide – just compare its structure with that of DDT (below) – but was found to have broad-spectrum anti-bacterial properties. Compounds with phenolic –OH groupings often have antibacterial and antiseptic properties, like Dettol, and organochlorine compounds frequently act as pesticides.
 |
 |
 |
Triclosan |
DDT |
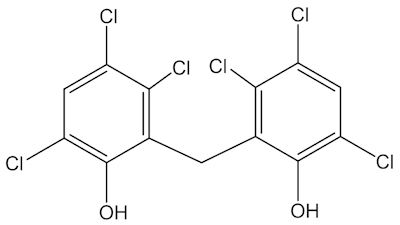
Hexachlorophene |
It replaced less pleasant molecules like hexachlorophene (below) in surgical scrubs from 1972, before finding other uses, not just in deodorant soaps, shower gels, handwashes, toothpastes and mouthwashes, but also in consumer products like carpets, kitchen tiles, children’s toys, athletic clothing and cutting boards.

Some consumer products which contain triclosan.
And triclocarban?
It is another anti-microbial compound, though not so widely used as triclosan.
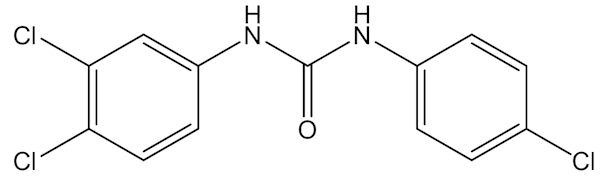
Triclocarban
How does triclosan work?
Triclosan interferes with lipid synthesis by inhibiting the enzyme enoyl-acyl carrier protein reductase (ENR) which makes the fatty acids (long-chain carboxylic acids), at what is known as the FabI step. This leads to weaker cell membranes in the bacterial cells, which then die. Humans do not have an ENR enzyme and thus are not affected.
So?
It has been argued that this might lead to resistance in bacterial cells to triclosan, and this could be a real worry, given the widespread use of triclosan.
 How effective is triclosan?
How effective is triclosan?
If you wash your hands with soap and water, studies suggest it could prevent up to 40% of diarrhoea and 25% of pneumonia cases. These are especially big killers of children in the Third World (where, sadly, few people wash their hands). The jury is divided on how effective triclosan is. Whilst triclosan certainly kills bacteria, studies have reported that ordinary soap works just as well as soaps with antibacterials in reducing infections, if you wash your hands properly. Of course, normal hand-washing removes the soap soon after it has been applied, which would only give the antibacterial agent a few seconds in which to work. A study in Pakistan found no significant difference in diarrhoea between households using antibacterial soap and those using plain soap. This may be because triclosan works against some gram-positive bacteria, but does not affect gram-negative bacteria, viruses, or parasites. (This is not particularly surprising - you do not expect antibacterial agents to work against viruses).
If it is so good, what’s the problem with triclosan?
The effectiveness of triclosan in applications like surgical scrubs is not disputed. The problem lies with all the extraneous applications of triclosan in “personal care” products. The U.S. state of Minnesota has legislated to ban the use of triclosan outside of clear medical uses as in hospitals; other states may follow suit. It looks as if some firms which make antibacterial soaps may phase out triclosan. The U.S. Food and Drug Administration (FDA) is to consider regulating the use of both triclosan and the similar triclocarban; they are concerned about how effective they are and also about reports that they are endocrine disruptors, as well as being worried about their contributing to bacterial resistance to antibiotics. There is a very real concern about the spread of triclosan in living systems. It is widely detected in breast milk, and a very recent trial showed it turning up in maternal urine and cord plasma. Some people worry that triclosan may affect viruses negatively, by triggering mutations that would make viral infections more, not less, likely.
Are there any other potential problems?
A significant proportion of triclosan makes it into waste water. This could be damaging to fish and marine invertebrates, whilst chlorine – a substance commonly used to make water safe to drink - reacts with triclosan forming chloroform, a potential carcinogen.
But what can replace triclosan?
The problem is partly one of overexposure of a chemical – you can have too much of a good thing. Scientists are now looking for a better product, something that is a very effective wide-spectrum antimicrobial chemical that is also not toxic to humans, to animals and the environment, something that does not bioaccumulate and biodegrades easily. Can there be such a “green” pharmaceutical? A number of alternative FabI inhibitors have been studied, with at least one (below) undergoing clinical trials.
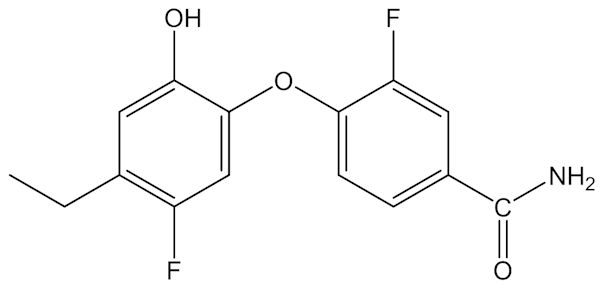
As yet unnamed molecule which might be an alternative to triclosan

Bibliography
- J. Kemsley, Chem. Eng. News, 92 (June 23 2014), 10-13 (overview: “Triclosan Under The Microscope”)
How triclosan works
- L. M. McMurry, M. Oethinger and S. B. Levy, Nature, (1998), 394, 531-532 (mechanism of action)
- C. W. Levy, A. Roujeinikova, S. Sedelnikova, P. J. Baker, A.R. Stuitje, A. R. Slabas, D. W. Rice and J. B. Rafferty, Nature, (1999), 398, 383-384. (structure of E. coli enoyl reductase-NAD1-triclosan complex)
- X. Qiu, C. A. Janson, R. I. Court, M. G. Smyth, D. J. Payne and S. S. Abdel-Meguid, Protein Science, (1999) 8 2529–2532. (structure of E. coli enoyl reductase-NAD1-triclosan complex)
- R. J. Heath J. R. Rubin, D. R. Holland, E. Zhang, M. E. Snow and C. O. Rock, J. Biol. Chem., (1999) 274, 11110–11114 (structure of FabI-NAD+-triclosan complex)
Effectiveness
- R. D. Jones, H. B. Jampani, J. L. Newman and A. S. Lee, Am. J. Infect. Control., (2000) 28, 184-196. (review of effectiveness and safety in health care settings)
- E. L. Larson, S. X. Lin, C. Gomez-Pichardo and P. Della-Latta, Ann. Intern. Med., (2004) 140, 321-329. (antibacterial products do not reduce the risk for primarily viral infections in households of healthy persons)
- S. P. Luby, M. Agboatwalla, J. Painter, A. Altaf, W. L. Billhimer and R.M. Hoekstra, JAMA, (2004) 291, 2547-2554 (study of soaps and diarrhoea in Pakistan)
- S. P Luby, M. Agboatwalla, D. R Feikin, J. Painter, W. Billhimer, A. Altaf and R. M Hoekstra, The Lancet, (2005) 366, 225-233. (antibacterial soap no more effective than plain soaps)
- K. L. Rule, V. R. Ebbett and P. J. Vikesland, Environ. Sci. Technol., (2005) 39 3176–3185 (triclosan reacts with chlorine to form chloroform)
- D. W. Schaffner, J. P. Bowman, D. J. English, G. E. Fischler, J. L. Fuls, J. F. Krowka and F. H. Kruszewski, J. Food Protection, (2014) 77 574-582. (triclosan reduces bacteria levels)
- K. Kupferschmidt, Science, (2014) 345, 1262-1263 (benefits of handwashing)
- R. U. Halden, Environ. Sci. Technol. (2014) 48 3603–3611 (review of antimicrobials)
- B. F. G. Pycke, L. A. Geer, M. Dalloul, O. Abulafia, A. M. Jenck and R. U. Halden, Environ. Sci. Technol., (2014) 48 8831−8838 (Foetal exposure to Triclosan and Triclocarban; presence in maternal urine and core blood plasma)
- A. D. Dayan, Food and Chemical Toxicology, (2007) 45 125–129 (triclosan in human breast milk)
Alternatives to triclosan
- V. Gerusz, A. Denis, F. Faivre, Y. Bonvin, M. Oxoby, S. Briet, G. LeFralliec, C. Oliveira, N. Desroy, C. Raymond, L. Peltier, F. Moreau, S. Escaich, V. Vongsouthi, S. Floquet, E. Drocourt, A. Walton, L. Prouvensier, M Saccomani, L. Durant, J.-M. Genevard, V. Sam-Sambo and C. Soulama-Mouze, J. Med. Chem., (2012) 55 9914−9928 (possible alternative antibacterials)
Scientific American on triclosan


 Back to Molecule of the Month page. [DOI:10.6084/m9.figshare.5259145]
Back to Molecule of the Month page. [DOI:10.6084/m9.figshare.5259145]

![]()
![]()
![]()
![]()






 How effective is triclosan?
How effective is triclosan?