![]()
![]()
Mike Thompson and Anthony Woodman
Winchester College, UK
![]()
Also available: Chime Enhanced, JMol, and VRML versions.
![]()
Uric acid is a colourless, odourless and tasteless solid with molecular formula C5H4N4O3. Its systematic name is 2,6,8-trioxypurine. Historically, it has been called lithic acid or 8-hydroxyxanthine. Safety data on uric acid is unremarkable. It is largely insoluble in water. Rather than melt, it decomposes in air above 250°C. Small amounts of uric acid are found in human urine as it is an excretory product at the end of nucleic acid catabolism (breakdown).
Commercially, uric acid has been prepared from guano by extracting it with alkali and then precipitating with acid. Guano is essentially the droppings of bats or seabirds, and has been found in large quantities on some islands. It even got a mention in the James Bond book 'Dr No' by Ian Fleming.

Adenine and guanine are probably two of the most famous purines as they are DNA bases. Figure 1 shows the similarity between caffeine and uric acid which are also purines. Recent research suggests that the brain may benefit from daily caffeine intakes - hardly surprising since it is also known that uric acid at low levels is implicated in certain neurological disorders. Higher levels of uric acid might be linked to longevity because of its strong antioxidant properties. In humans, about half of the blood's anti-oxidative capacity is believed to be due to uric acid.
Low levels of uric acid in blood serum have also been linked to multiple sclerosis (MS). Conversely, high levels of uric acid have been associated with gout. Interestingly, a study of the medical records of 20 million U.K. patients showed that MS and gout occur almost entirely mutually exclusively. I know what I'd rather have…
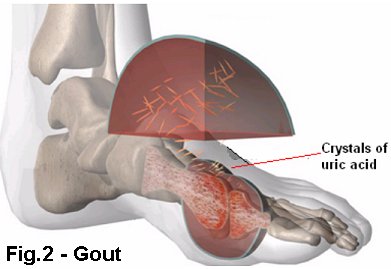 Gout
GoutGout is a painful condition, and a form of arthritis, that causes inflammation of the joints. This happens more often in men over 40 and affects about 0.5% of the population. Gout has been recognised for over 2500 years since the time of the Greek physician Hippocrates. Gout was considered to be caused by over-indulgence in food and alcohol, as it mostly afflicted the wealthy. It has been found that eating herring, sardines, heart or mussels may increase serum levels of uric acid. Famous gout sufferers have included; Alexander the Great, Henry VIII, Christopher Columbus, Leonardo da Vinci, Isaac Newton, John Milton and Alfred Lord Tennyson. Gout is usually associated with hyperuricemia - elevated uric acid levels in blood serum. Gout is caused by the deposition of sodium urate (the salt of uric acid) crystals in the joints. The pain and inflammation is often found in the joint of the big toes as seen in Figure 2.
There are several detection methods for uric acid. A new method was reported in 2006 by the reported by the British Scientist David Parker which utilizes luminescent lanthanide complexes.
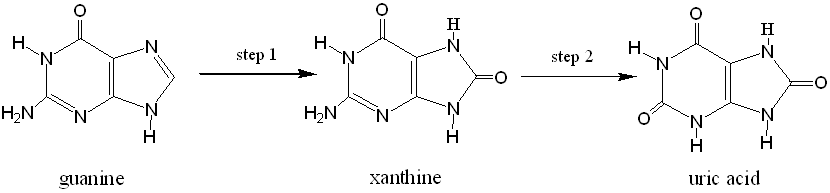
Figure 3
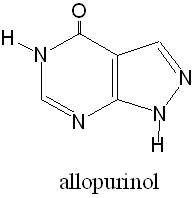 Figure 3 shows how one of the four DNA bases, guanine, is metabolised into uric acid via the intermediate xanthine.
Figure 3 shows how one of the four DNA bases, guanine, is metabolised into uric acid via the intermediate xanthine.
It is possible to treat gout with allopurinol, which works by inhibiting xanthine oxidase, the enzyme that produces uric acid from xanthine in step 2. The drug competes for the active site with xanthine; taking 100-300 mg each day of allopurinol usually works well to prevent gout attacks by reducing uric acid levels in the blood.
Insects produce uric acid during protein breakdown (catabolism). The uric acid is not just excreted and has found some interesting uses. Uric acid helps to make butterflies iridescent. Its crystals also reflect the light produced in fireflies by bioluminescence in a similar way to mirrors in a car headlight.
|
|

 The end of the metabolic pathway for most mammals is not uric acid but allantoin. This final transformation (step 3 in Figure 4) takes place in the liver catalyzed by the enzyme urate oxidase. Humans, some primates and - curiously enough - dalmations are one of the few mammals that do not possess this enzyme and thus cannot oxidise uric acid to allantoin.
The end of the metabolic pathway for most mammals is not uric acid but allantoin. This final transformation (step 3 in Figure 4) takes place in the liver catalyzed by the enzyme urate oxidase. Humans, some primates and - curiously enough - dalmations are one of the few mammals that do not possess this enzyme and thus cannot oxidise uric acid to allantoin.
 |
Click either puppy for an optical illusion. |
 Uric acid in the desert
Uric acid in the desertThe kangaroo rat (photo, right) is adapted to very dry environments. It is able to conserve water by producing uric acid instead of urea as an excretory product, and is therefore calssified as a uricotelic organism (as are birds and repitiles). This means that a complex metabolic pathway converts all amino acids to three other amino acids; glycine, glutamine or aspartic acid. These amino acids combine and are then degraded into uric acid. The payback for this costly operation is the conservation of water, which is a useful piece of evolution for a desert dweller like the Kangaroo rat.
A wide variety of organisms make use of uric acid to produce pigments called pterins, e.g. xanthopterin, erythropterin and drosopterin. This class of molecules was first isolated from butterfly wings and was named from the Greek word pteron, meaning 'wing'. A whole range of colours - a result of conjugation around the aromatic ring systems - have been achieved by subtle changes to the rings and the functional groups. An interesting article was written on this subject entitled "Insects, such as butterflies, wasps and 'true bugs' (heteroptera), cold-blooded vertebrates and even crustaceans do this" in Nature: March 4th 1950, 165, 367-8.
| |
| |
|
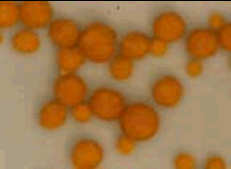 Uric acid (kidney) stones
Uric acid (kidney) stones Elevated blood uric acid levels can also cause kidney disease. In a few people the uric acid crystals form kidney stones (photo, right). Allopurinol can be used to prevent uric acid kidney stones as well as gout.
Uric acid is combined in the form of urate of ammonia, and is the chief constituent of the urine of birds and reptiles, forming the white part. This helps to keep the birds' weight down which is necessary for flight. Beware, it can and does damage paint on cars as it is acidic.


![]()