
![]()
VANILLIN
The flavour of vanilla icecream
![]()
Simon Cotton
Uppingham School, Rutland, UK
![]()
Molecule of the Month February 2008
Also available: JSMol version.
![]()

 |
VANILLINThe flavour of vanilla icecream
Simon Cotton
Molecule of the Month February 2008
|
 |
Not so simple, and not just because vanilla grows on vines, not trees. The freshly harvested green vanilla pods do not smell "vanilla" as the vanillin is locked up inside them as a β-D-glycoside (see below).
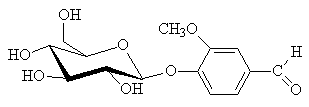 |
| The glucoside containing vanillin |
The pods have to be cured, during which time they become deep brown, and enzymes release the vanillin from the glycoside, along with over a hundred other molecules, all of which contribute to the authentic vanilla aroma. Vanillin itself makes up about 2% of the final mass of the "cured" beans.


Vanilla Beans..... ...and vanilla pods.
 And it is a modern flavouring?
And it is a modern flavouring?Not so. The vanilla plant, Vanilla planifola (see photo at the top right of the page), originates in subtropical forests in Mexico and parts of Central America, and it was the Mayan and Aztec civilisations which first realised the potential of vanilla, using it to lighten up the flavour of the chocolate that they drank. The Aztec leader Moctezuma hinted to HernŠn Cortťs (who led the Spanish invaders) that the Aztecs' vanilla flavoured chocolatl drinks were a very useful aphrodisiac. After their conquest of these areas, the Spaniards brought vanilla to Western Europe in the early 16th century.
People discovered lots of other uses, as a sweetener and flavour enhancer, in food flavourings and confectionary, not just chocolate, in medicines and cleaning products (including soap), not to mention masking flavours in animal feed. Eventually it found its way into Coca-Cola, and into some great perfumes and fragrances. Nowadays it is widely used in chemical synthesis, for example in the manufacture of L-dopa, for treatment of Parkinson's disease, of the antibacterial compound trimethoprin, and of the heart drug papaverin. In research laboratories, acidic solutions of vanillin in ethanol are used as a developing (visualising) agent for t.l.c. plates as it gives coloured spots with a range of different compounds.
Production of vanilla spread well beyond Central America, once it was discovered that vanilla orchids could be fertilised manually. It has been grown in tropical regions as diverse as the Caribbean islands, Tahiti, Rťunion, Madagascar (the world leader) and the Indonesian archipelago. Despite this, since the 19th century, demand has vastly outstripped production of natural vanilla, so that synthetic routes to vanillin had to be found. Nowadays less than 5% of vanilla flavouring comes from natural sources; many people are so conditioned to the use of synthetic "vanilla" flavour that they prefer it to the real thing, and may wonder about the tiny flakes of vanilla beans they find in an up-market ice cream.
Because genuine vanilla extract from pods is up to 200 times more expensive than counterfeit vanilla essence using synthetic vanillin, detection of fake vanilla is important. Some fakes can be detected using chromatography, but the goalposts keep moving. One of the first detection methods relied on the fact that vanillin obtained from fresh vanilla pods had higher radiocarbon (14C) levels than vanillin from petrochemical sources (though it did not distinguish it from material obtained from fresh lignin); in response, the fakers added extra 14C to their vanillin. Analysts then examined the 13C/12C ratios in the samples by mass spectroscopy. A distinction is possible because the vanilla orchid has an unusual biosynthetic pathway for vanillin so that more 13C than usual is incorporated; "fake" vanilla extract will originate in material with a lower 13C/12C ratio; in their turn, the fakers added 13C to their molecules, usually just at one site - analysts have hit back by using site-sensitive NMR techniques (SNIF-NMR). On some levels, this problem is somewhat semantic, since the two products are identical by taste.
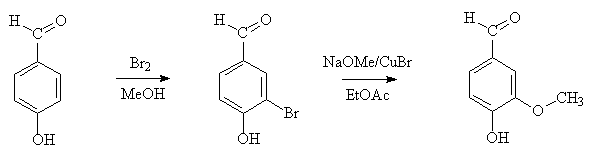
If you just want to make a few grams, a very convenient laboratory synthesis has recently been described, involving electrophilic bromination of 4-hydroxybenzaldehyde, followed by copper-catalysed methoxylation.
As far as large-scale industrial syntheses go, a classic early method starts from eugenol, which occurs naturally in cloves, nutmeg and cinnamon. This isomerises to isoeugenol in alkaline solution, and this in turn can be oxidised (by nitrobenzene) to vanillin.
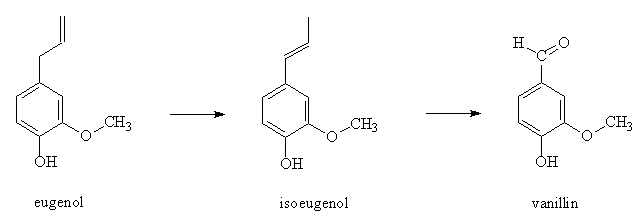
Other oxidising agents like acidified potassium dichromate can be used, but then the OH group has to be protected by acetylation prior to oxidation.
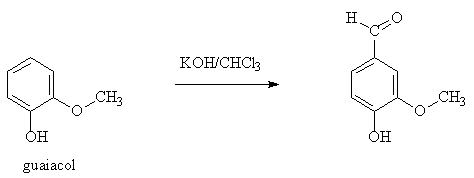
More dramatic is the Reimer-Tiemann method (1876). Here another natural product, guaiacol, obtained from sources like pine tar, gains an aldehyde group when refluxed with an alkaline solution of chloroform. The mechanism is now known to involve the carbene species :CCl2.
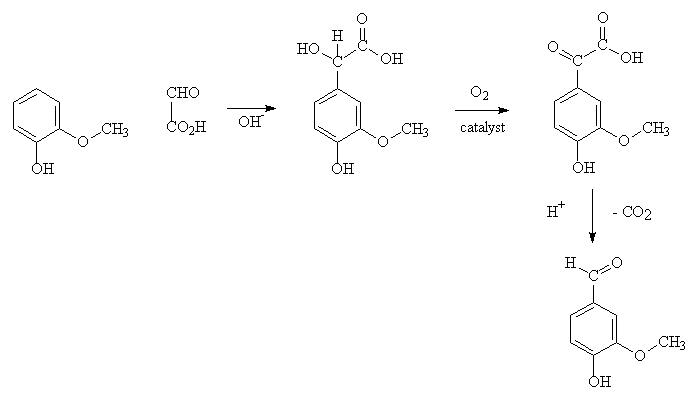
A more modern method reacts guaiacol, itself obtained from catechol, with glyoxylic acid, and that is how a lot of vanillin is produced today.
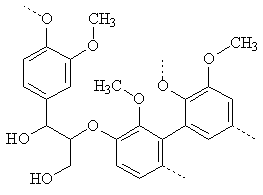
Early in the 20th century, chemists found out that lignin was a source of vanillin; lignin makes up around 5% of the sulphite waste waters from paper mills. Lignin is a polymer with a strengthening role in woods and in the cell walls of plants (simplified structure, below). On combustion of wood at relatively low temperatures, the polymeric structure breaks down into small units, and small molecules such as vanillin and guiaicol are formed; these are responsible for the smell and taste of smoked food. Wine stored in oak casks similarly gains some of its flavour from leached vanillin. From the 1920s, much of the world's vanillin was extracted from lignin waste.
 |
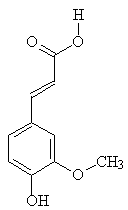 |
| Simplified structure of lignin | Ferulic acid |
Not only did this process make vanillin, it also cut down on organic discharges, but at present this is out of favour in North America, owing to environmental concerns, as over 160 tons of "caustic waste" are produced per ton of vanillin. Ferulic acid (structure, above) found in many plants, is another source of vanillin, via microbial routes.
 A new lignin route to vanillin is a possibility, ever since Dr Mayu Yamamoto, of the International Medical Centre of Japan, realised that cow dung contained a lot of lignin, from the animal's feed. She found a way of extracting the lignin, then converting this to vanillin. In fact, many herbivores' excrements yield vanillin. Her achievement saw her win the 'Ig Nobel' prize for Chemistry in 2007, the award - a send-up of the "real" Nobel prize - being presented at a ceremony at Harvard University on October 4th 2007. At the time, a leading ice-cream parlour in Cambridge, Massachusetts (home of Harvard) created a new ice cream flavour, "Yum-A-Moto Vanilla Twist", offering free public tastings. This was created from genuine vanilla beans; since food labelling laws will require the origin of vanillin to be disclosed, such vanillin of animal origin would only find non-food outlets.
A new lignin route to vanillin is a possibility, ever since Dr Mayu Yamamoto, of the International Medical Centre of Japan, realised that cow dung contained a lot of lignin, from the animal's feed. She found a way of extracting the lignin, then converting this to vanillin. In fact, many herbivores' excrements yield vanillin. Her achievement saw her win the 'Ig Nobel' prize for Chemistry in 2007, the award - a send-up of the "real" Nobel prize - being presented at a ceremony at Harvard University on October 4th 2007. At the time, a leading ice-cream parlour in Cambridge, Massachusetts (home of Harvard) created a new ice cream flavour, "Yum-A-Moto Vanilla Twist", offering free public tastings. This was created from genuine vanilla beans; since food labelling laws will require the origin of vanillin to be disclosed, such vanillin of animal origin would only find non-food outlets.
Vanillin came into the picture in the debate over the dating of the Shroud of Turin, which many believe to be the burial cloth of Jesus Christ. Radiocarbon datings made in 1988 suggested that the age of the Shroud was 1260-1390 AD, at odds with chemical evidence. Recent examination of vanillin levels in the Shroud showed vanillin to be present in cloth added following a fire in 1532, but absent from the older parts of the Shroud. Vanillin levels in material containing lignin fall with time, thus it cannot be detected on the 2000-year-old Dead Sea Scrolls linen, either. Examination of the kinetics of vanillin loss indicated the main part of the Shroud to be between 1300 and 3000 years old, suggesting that the radiocarbon dating sample may have been taken from a newer piece of material.
Two classic Guerlain perfumes use vanillin molecules as key elements. In 1889, Aimť Guerlain created Jicky, which over a century later remains amazingly contemporary. It is often said to be the first "modern" perfume, utilising synthetic ingredients. The initial impact is provided by top notes of lavender, lemon and bergamot, followed by a similarly cold and rosy middle, supported by a warm base of amber, musk and, above all, vanilla ("un ballet olfactif").
 |
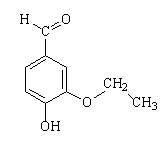 Ethyl-vanillin |
 |
 Aimť Guerlain's nephew Jacques was responsible for Shalimar in 1925 (named after the garden that the Mughal emperor Shah Jahan created for his wife). It is said that he added synthetic ethyl vanillin to a sample of Jicky. Ethyl vanillin (structure shown in the middle, above), which does not occur naturally, has a much stronger vanilla smell than its methyl analogue, vanillin itself. The odour threshold of ethyl vanillin is given as 0.000007 mg/m3 of air, in comparison for a value for vanillin itself in the range 0.005 - 0.000001 mg/m3 of air. In Shalimar, the lemon and bergamot impact again presages the floral middle passage (rose, jasmin) against a warm vanilla base. The result is quite sensual. Just think of Mireille Darc (photo, right), a French actress famous for wearing Shalimar, in Les Coeurs BrŻlťs.
Aimť Guerlain's nephew Jacques was responsible for Shalimar in 1925 (named after the garden that the Mughal emperor Shah Jahan created for his wife). It is said that he added synthetic ethyl vanillin to a sample of Jicky. Ethyl vanillin (structure shown in the middle, above), which does not occur naturally, has a much stronger vanilla smell than its methyl analogue, vanillin itself. The odour threshold of ethyl vanillin is given as 0.000007 mg/m3 of air, in comparison for a value for vanillin itself in the range 0.005 - 0.000001 mg/m3 of air. In Shalimar, the lemon and bergamot impact again presages the floral middle passage (rose, jasmin) against a warm vanilla base. The result is quite sensual. Just think of Mireille Darc (photo, right), a French actress famous for wearing Shalimar, in Les Coeurs BrŻlťs.
![]()
![]()
![]() Back to Molecule of the Month page. [DOI:10.6084/m9.figshare.5249122]
Back to Molecule of the Month page. [DOI:10.6084/m9.figshare.5249122]