Atmospheric Pressure Chemical Ionisation (APCI)
Atmospheric pressure chemical ionisation (APCI) is an analogous
ionisation method to chemical ionisation (CI). The significant
difference is that APCI occurs at atmospheric pressure and has its
primary applications in the areas of ionisation of low mass
pharmaceutical compounds (APCI is not suitable for the analysis of
thermally labile compounds). The general source set-up (see figure 1)
shares a strong resemblance to electrospray ionisation (ESI) and as
such is most commonly used in conjunction with HPLC or other flow
separation techniques. Where APCI differs to ESI, is in the way
ionisation occurs. In ESI, ionisation is bought about through the
potential difference between the spray needle and the cone along with
rapid but gentle desolvation. In APCI, the analyte solution is
introduced into a pneumatic nebulizer and desolvated in a heated
quartz tube before interacting with the corona discharge creating
ions.
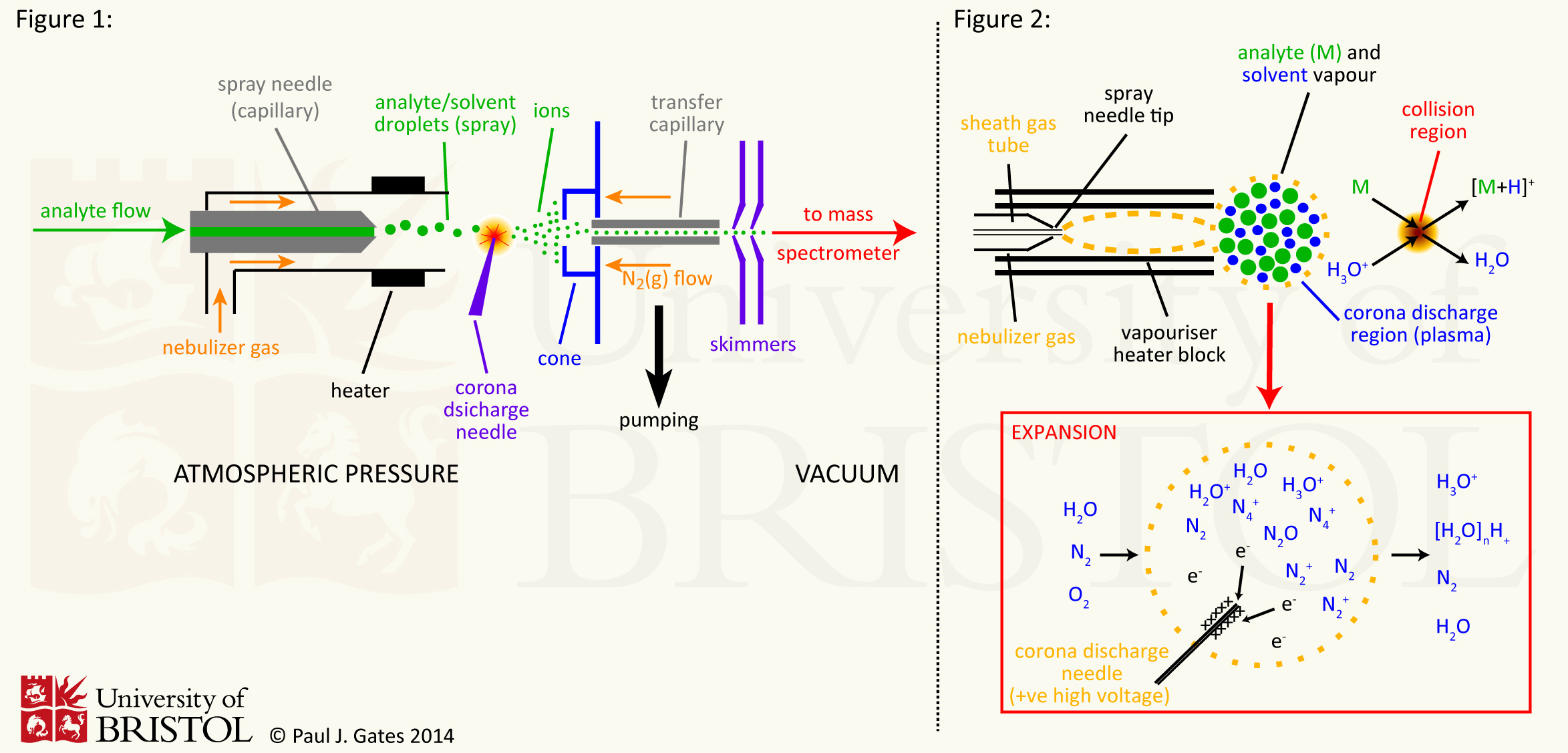
The corona discharge replaces the electron filament in CI - the
atmospheric pressure would quickly "burn out" any filaments - and
produces primary N2°+ and N4°+
by electron ionisation. These primary ions collide with the vaporised
solvent molecules to form secondary reactant gas ions - e.g. H3O+
and (H2O)nH+ (see figure 2). These
reactant gas ions then undergo repeated collisions with the analyte
resulting in the formation of analyte ions. The high frequency of
collisions results in a high ionisation efficiency and thermalisation
of the analyte ions. This results in spectra of predominantly
molecular species and adduct ions with very little fragmentation. Once
the ions are formed, they enter the pumping and focussing stage in
much the same as the other atmospheric pressure ionisation sources
(for example ESI).

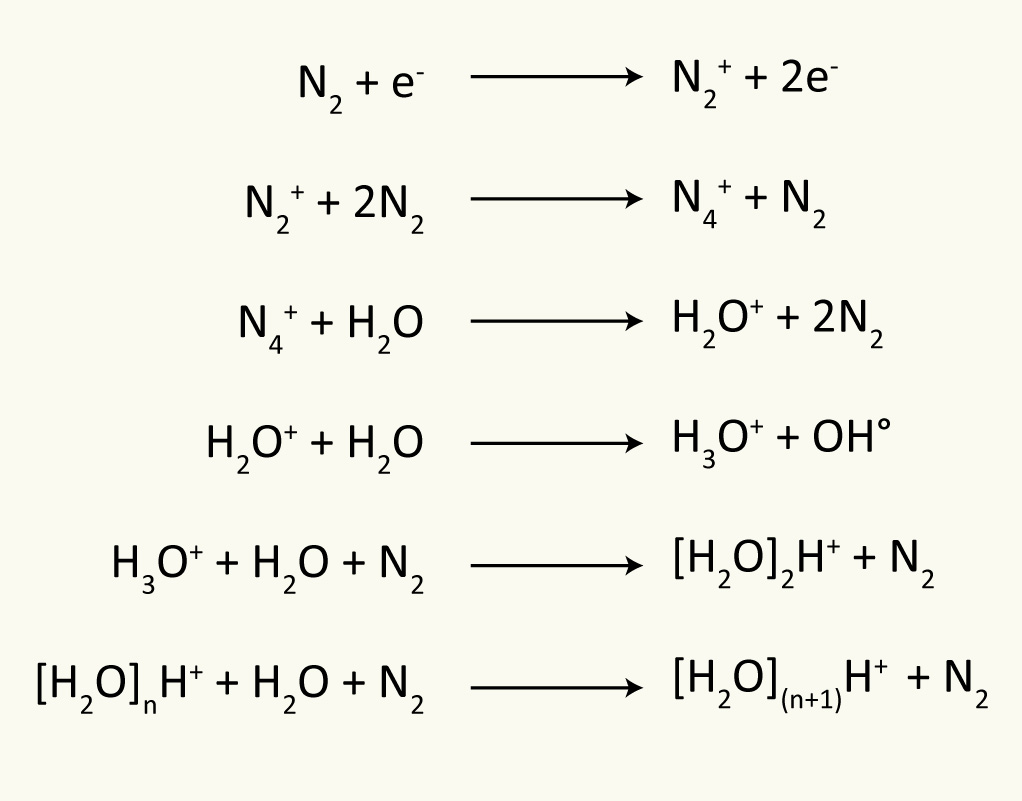
| Reactions in the plasma region: Assuming nitrogen is the sheath and nebulizer gas with
atmospheric water vapour present in the source, then the
type of primary and secondary reactions that occur in the
corona discharge (plasma) region during APCI are as shown
in the scheme. The most abundant secondary cluster ion is
(H2O)2H+ along with
significant amounts (H2O)3H+
and H3O+. The reactions listed above
are ways to account for the formation of these ions during
the plasma stage. The protonated analyte ions are then
formed by gas-phase ion-molecule reactions of these
charged cluster ions with the analyte molecules. This
results in the abundant formation of [M+H]+
ions.
|
 |