Chemical Ionisation (CI)
Chemical Ionisation is the second of the techniques to be
considered a 'classical' ionisation method. It is still widely use
today for the analysis of low-mass, volatile, thermally stable organic
compounds especially when coupled with gas chromatography (GC-MS).
Chemical ionisation is a lower energy alternative to EI for volatile analytes. In CI, there is a reagent gas (usually ammonia or methane) in the ion chamber. The scheme below shows ion formation in CI using methane as the reagent gas. In the top equation methane is ionised by an electron beam in the same way as with EI. The ionised methane gas then reacts with un-ionised reagent gas to form the methanium carbocation (protonated methane). This step requires the CI reagent gas to be at a critical pressure - too low a pressure, and no ionisation of the analyte can take place. Methanium can then udergo proton transfer to the analyte [M] to form the protonated analyte molecule [M+H]+. If the pressure of the reagent gas is too high, then the side reactions can also occur, leading to formation of the analyte adduct ion - this is seen as an [M+C2H5] peak in the spectrum (occurring 28 m/z higher than the [M+H]+).
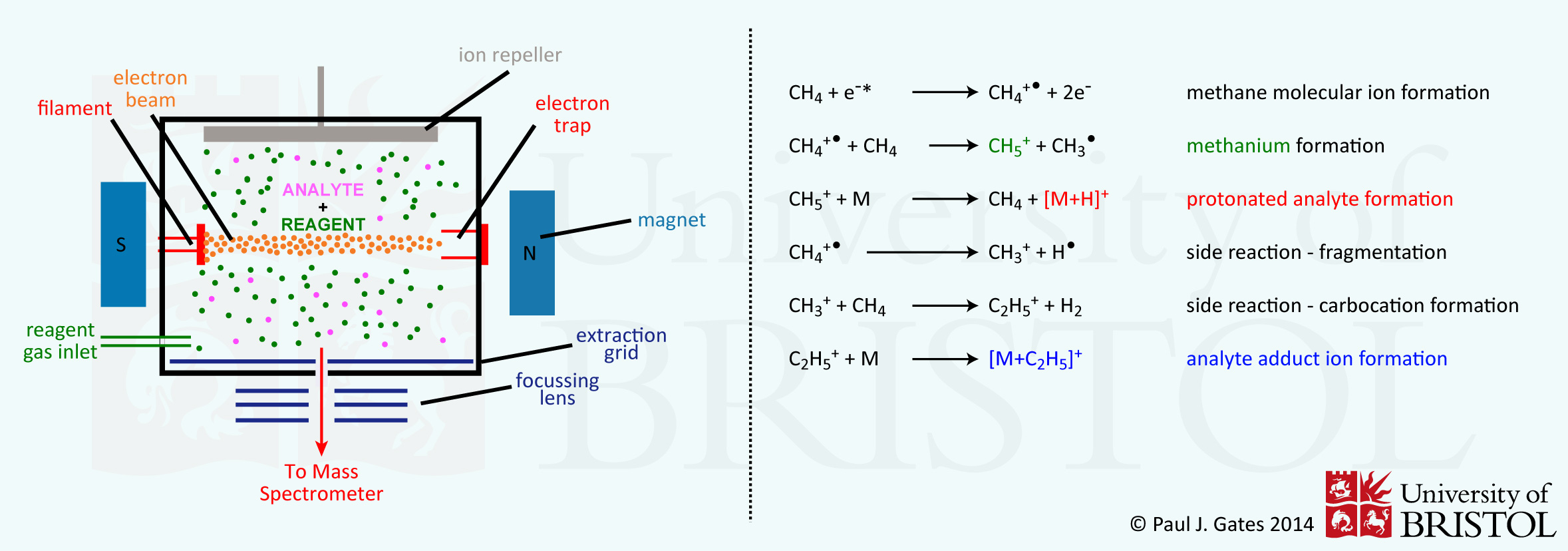
In CI, ionisation is due to proton transfer and is therefore a much lower energy process. This results in less residual energy being possessed by the protonated molecules so that fragmentation is greatly reduced. However, CI still requires volatilisation of the analyte, so thermal degradation of the analyte can still lead to fragment ions being observed. However, CI is generally considered a much 'softer' ionisation technique to EI, and until the development of the desorption/ionisation methods in the 1970's, was the only way to analyse most small, biologically important molecules (sugars, amino acids, lipids etc.).
Chemical ionisation is a lower energy alternative to EI for volatile analytes. In CI, there is a reagent gas (usually ammonia or methane) in the ion chamber. The scheme below shows ion formation in CI using methane as the reagent gas. In the top equation methane is ionised by an electron beam in the same way as with EI. The ionised methane gas then reacts with un-ionised reagent gas to form the methanium carbocation (protonated methane). This step requires the CI reagent gas to be at a critical pressure - too low a pressure, and no ionisation of the analyte can take place. Methanium can then udergo proton transfer to the analyte [M] to form the protonated analyte molecule [M+H]+. If the pressure of the reagent gas is too high, then the side reactions can also occur, leading to formation of the analyte adduct ion - this is seen as an [M+C2H5] peak in the spectrum (occurring 28 m/z higher than the [M+H]+).

In CI, ionisation is due to proton transfer and is therefore a much lower energy process. This results in less residual energy being possessed by the protonated molecules so that fragmentation is greatly reduced. However, CI still requires volatilisation of the analyte, so thermal degradation of the analyte can still lead to fragment ions being observed. However, CI is generally considered a much 'softer' ionisation technique to EI, and until the development of the desorption/ionisation methods in the 1970's, was the only way to analyse most small, biologically important molecules (sugars, amino acids, lipids etc.).