Quadrupole Ion Trap (QIT)
The quadrupole ion trap (QIT) mass analyser was developed in
parallel with the quadrupole mass analyser by the Nobel prize winning
mass spectrometry pioneer, Wolfgang Paul [1,2]. His work in the early
1950's lead to the development of the basic parameters of today's
benchtop instruments, however it took breakthroughs in design at
Finnigan MAT in the 1980's [3] to make the QIT-MS the simple to use
practical instrument it is today. QIT instruments are commonly used
and are very amenable to being coupled to ESI sources and liquid
chromatography systems.
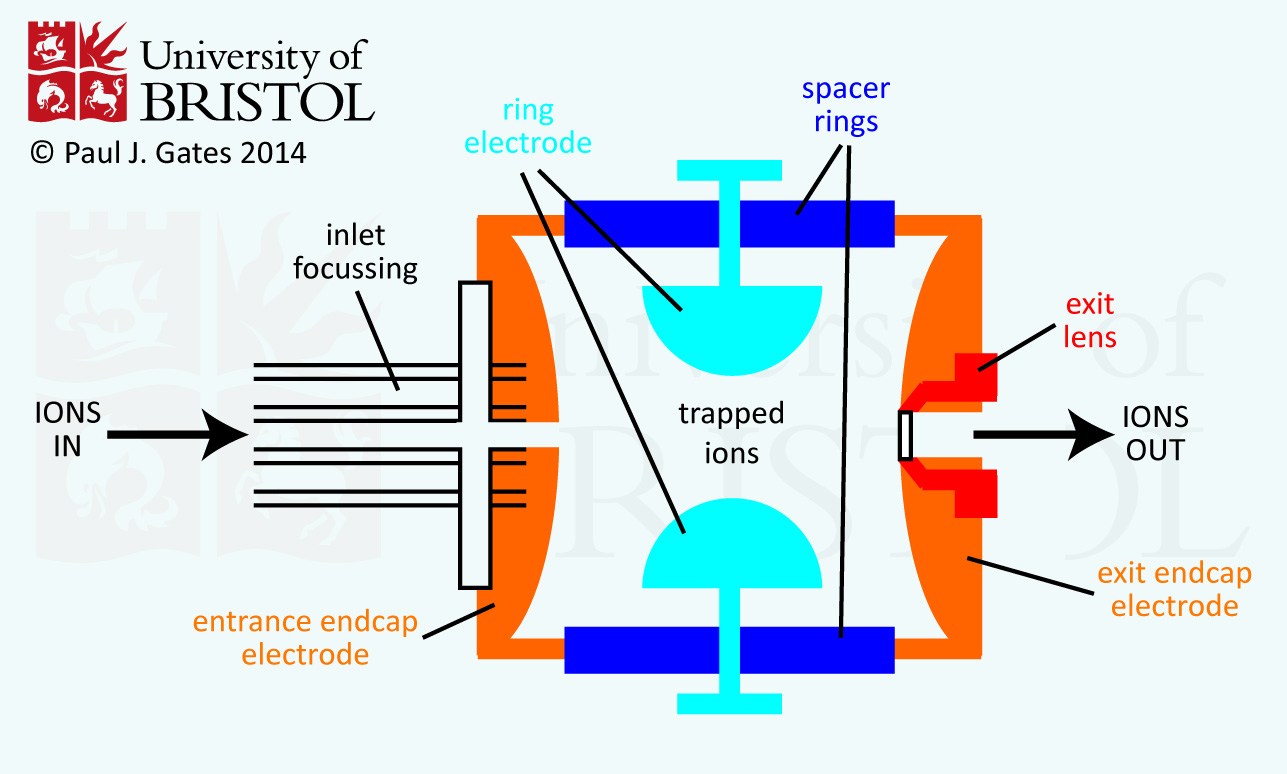
A schematic of the basic set up of a QIT mass analyser is shown in the figure. The ions, produced in the source of the instrument, enter into the trap through the inlet and are trapped through action of the three hyperbolic electrodes: the ring electrode and the entrance and exit endcap electrodes. Various voltages are applied to these electrodes which results in the formation of a cavity in which ions are trapped. The ring electrode RF potential (an AC potential of constant frequency but variable amplitude) produces a 3D quadrupolar potential field within the trap. This traps the ions in a stable oscillating trajectory. The exact motion of the ions is dependent on the voltages applied and their individual mass-to-charge (m/z) ratios. For detection of the ions, the potentials are altered to destabilise the ions resulting in their ejection through the exit endcap. The ions are usually ejected in order of increasing m/z by a gradual change in the potentials. This 'stream' of ions is focussed onto the detector of the instrument to produce the mass spectrum.
The very nature of trapping and ejection makes a quadrupolar ion trap especially suited to performing sequential mass spectrometry (MSn) experiments for structural based studies. It is possible to selectively isolate a particular m/z in the trap by ejecting all the other ions from the trap. Fragmentation of this isolated precursor ion can then be induced through collisional induced dissociation (CID). The isolation and fragmentation steps can be repeated a number or times and is only limited by the trapping efficiency of the instrument. MS5 experiments are fairly routine with this set-up as is the coupling of liquid chromatography to perform LC-MSn studies.
[2] W. Paul; Agewandte Chemie - International Edition, 29; 1990, p739.
[3] G. C. Stafford et al.; International Journal of Mass Spectrometry and Ion Processes, 60; 1984, p85 and Analytical Chemistry, 59; 1987, p1677.
A schematic of the basic set up of a QIT mass analyser is shown in the figure. The ions, produced in the source of the instrument, enter into the trap through the inlet and are trapped through action of the three hyperbolic electrodes: the ring electrode and the entrance and exit endcap electrodes. Various voltages are applied to these electrodes which results in the formation of a cavity in which ions are trapped. The ring electrode RF potential (an AC potential of constant frequency but variable amplitude) produces a 3D quadrupolar potential field within the trap. This traps the ions in a stable oscillating trajectory. The exact motion of the ions is dependent on the voltages applied and their individual mass-to-charge (m/z) ratios. For detection of the ions, the potentials are altered to destabilise the ions resulting in their ejection through the exit endcap. The ions are usually ejected in order of increasing m/z by a gradual change in the potentials. This 'stream' of ions is focussed onto the detector of the instrument to produce the mass spectrum.

The very nature of trapping and ejection makes a quadrupolar ion trap especially suited to performing sequential mass spectrometry (MSn) experiments for structural based studies. It is possible to selectively isolate a particular m/z in the trap by ejecting all the other ions from the trap. Fragmentation of this isolated precursor ion can then be induced through collisional induced dissociation (CID). The isolation and fragmentation steps can be repeated a number or times and is only limited by the trapping efficiency of the instrument. MS5 experiments are fairly routine with this set-up as is the coupling of liquid chromatography to perform LC-MSn studies.
References:
[1] W. Paul & H. Steinwedel; Zeitschrift für Naturforschung, 8A; 1953, p448.[2] W. Paul; Agewandte Chemie - International Edition, 29; 1990, p739.
[3] G. C. Stafford et al.; International Journal of Mass Spectrometry and Ion Processes, 60; 1984, p85 and Analytical Chemistry, 59; 1987, p1677.