Cyclic Conjugated Polyenes
The ‘Circle’ Method; Benzene and Aromaticity
See Atkins page 438,
and Carroll
‘Perspectives on Structure and Mechanism in Organic Chemistry’ pgs. 205-208
It
is easy to write down the secular equations and secular determinant for
benzene.
We number the carbon atoms as below:
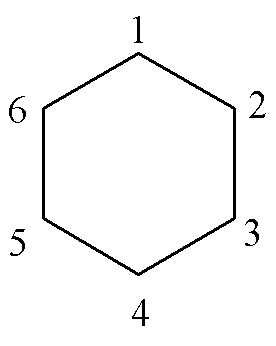
Consider
atom No. 1. It is next to atoms 6 and
2; so the corresponding secular equation is:
xc1 + c2 + 0´c3 + 0´c4 + 0´c5+ c6 = 0
or
xc1 + c2 + 0 + 0 + 0 + c6
= 0
· Each secular
equation involves just 3 non-zero terms.
· All the six secular
equations have the same structure, and they can be obtained from each other by
permuting the indices (i.e. by just adding one to the indices of the preceding
equation).
· The equations
can be solved analytically making use of trigonometric functions.
· These
trigonometric solutions may in turn, rigorously, be represented by a simple
geometric construction- this is the ‘Circle method’