Aromaticity
From
the results of the Circle Method, we find that we get a closed shell, stable
system only for cyclic polyenes with (4n+2) p electrons (n=0,1,2…).
Such systems have the highest
delocalization energies and are said to be “aromatic”.
The classic example is benzene:
p electron
energy = 2(a+2b) + 4(a+b) = 6a+8b
Compare to equivalent, non-delocalized model:
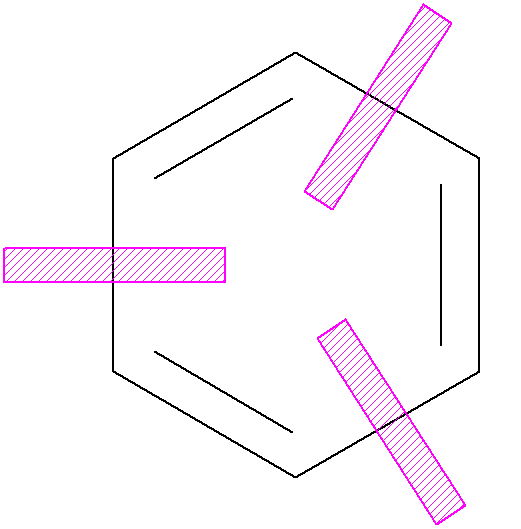
delocalization energy = p electron energy - energy of 3 isolated double bonds
= 6a+8b - 3(2a+2b)
= 2b
» –150 kJ mol-1
This is the energy by which the delocalized p electrons in
benzene are more stable than those in 3 isolated double bonds.