The
delocalization energy of 1,3-butadiene
The
delocalization energy is the extra stabilization that comes from letting the
electrons spread over the whole molecule: each molecular orbital spreads
further than just one pair of atoms.
To calculate how much stabilization
this provides, in other words to calculate the delocalization energy, we
compare the energy of the real molecule with one in which the electrons can’t
spread out.
Non-delocalized 1,3-butadiene (with a ‘wall’
between the double bonds):
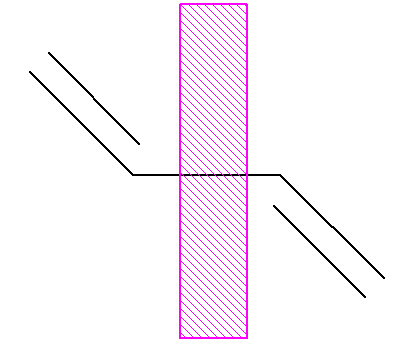
To find the delocalization energy, first
calculate the total p-electron energy of (real) butadiene:
p electron energy of 1,3
butadiene
= 2(a+1.62b) + 2(a+0.62b)
= 4a + 4.48b
Then calculate the energy of the same number
of p electrons in isolated (non-delocalized) bonds (i.e. can
think of this as butadiene with a ‘wall’ to stop the p electrons spreading):
p electron energy of an
equivalent number of isolated double bonds
= 2´(p electron
energy of ethene)
|
For
butadiene: p-electron energy of an equivalent
number of double bonds |
= |
2´(p electron
energy of ethene) |
= 2´(2a+2b)
= 4a+4b
Therefore the
delocalization energy of butadiene
= (4a + 4.48b) – (4a+4b)
= 0.48b
Delocalization
energy of allyl (C3H5)
- In the allyl
radical, there are 3 p electrons.
· 3 carbon 2p orbitals form 3 p molecular orbitals
- The energies
of the three molecular orbitals are (a + b√2), a and (a – b√2) (bonding, nonbonding and antibonding)
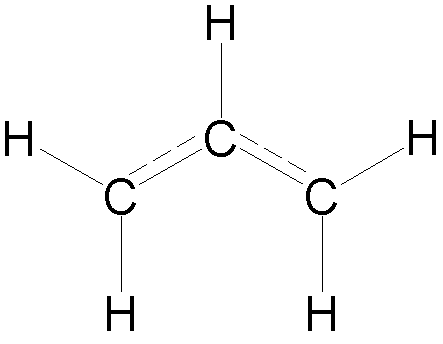
The total p electron
energy of allyl radical is therefore:
2´(a + b√2) +
a
= 3a + 2b√2
3a + 2.83b
In a model allyl radical with a wall to stop the p electrons delocalizing:
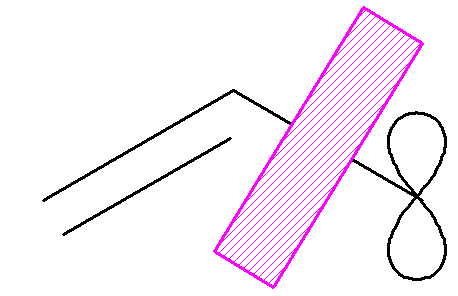
the total p electron energy
is
2´(a + b) + a = 3a +2b
(remember a is the
energy of an electron in a carbon 2p orbital – you need to add a for the energy of the odd electron)
So the delocalization energy of allyl radical is
3a + 2.83b – 3a +2b
= 0.83b