Observations of nanotube and ‘celery’ structures following diamond CVD on single crystal diamond substrates
This work was carried out in collaboration with M. Baines of the University of Bristol Physics Department.
1. Introduction
Following the preparation of macro quantities of C60 by Kratschmer et al [[1]] a number of different forms of carbon have been synthesised leading to interest in such materials for use in electronics applications. Since Iijima reported the preparation of carbon nanotubes (CNTs) [[2]], methods such as high pressure arc [[3]], laser ablation [[4]] and chemical vapour deposition (CVD) [[5],[6],[7]] have been developed to produce such material. The resulting CNTs have the structure of a rolled up graphite sheet, capped at each end with a carbon dome.
Similarly, carbon nanoparticles (CNPs) have been prepared by microwave plasma CVD, using growth conditions similar to those for CNTs, i.e. ~5%CH4/H2, 30 Torr, substrate temperature ~600°C and growth time of 15 minutes, with both methods requiring a metal catalyst (usually Ni) for growth. Other techniques, such as hot filament CVD [[8]] and cathodic arc [[9]] deposition, have also been used to produce CNPs.
Multi channel carbon tubules have been synthesised by Mukhopadhyay et al [[10]]. In their work camphor was pyrolysed and the soot was vacuum deposited resulting in multichannel-multilayer tubules of the order of 1000 mm long, with diameters of ~10 mm. The structure of these tubules was reported to differ from that of standard CNTs by being neither that of diamond, nor graphite. The same group have gone on to produce so-called spongy carbon ‘nanobeads’ by a method in which Ar was flowed through a camphor/ferrocene mixture at 50°C, and into a quartz tube at 1000°C [[11]]. The resulting beads were hollow and amorphous in structure, with diameters in the range of 0.2-0.8 mm, and are reported to form chains covered in a graphitic shell with a fibrous outer structure.
We now present observations of whisker formations found on the surface of diamond films that have been grown homoepitaxially by microwave CVD. These structures seem to be of two types, with similarities to both the carbon tubules and nanobeads discussed above, although they have a ‘celery-like’ structure that has not been hitherto reported.
2. Experimental
Deposition experiments were preformed using an ASTeX-style 2.45 GHz microwave plasma CVD reactor. Single crystal (100) HTHP diamond substrates (Sumitomo) were placed on an alumina plate on a cooled substrate holder. The substrates were thus elevated about 1 mm into the plasma, enabling automatic heating of the substrate to ~900°C (as measured by a two-colour optical pyrometer). The chamber pressure was 40 Torr with an applied microwave power of 1 kW, with deposition lasting 8 hours. All of the films were exposed to a hydrogen plasma (10 minute) after growth, and were then cooled back to room temperature in flowing H2. The feedstock gases used were H2 (99.999% purity), CH4 (99.999% purity), with a total gas flow of 200 sccm and gas mixing ratio of 1%CH4/H2.
Films and whiskers were examined by scanning electron microscopy (SEM) and transition electron microscopy (TEM). TEM samples were prepared by placing the substrate face down onto a mesh grid with filter paper underneath. Using a pipette, a few drops of ethanol were gently added. The ethanol was drawn into the filter paper causing some of the growth structures to become lodged on the grid. After allowing the components to dry, the substrate was removed, and a second grid attached on top of the first, effectively sandwiching material between them. Selected area diffraction was also utilised to investigate the crystallinity of the whiskers. The whiskers were also analysed by energy dispersive X-ray analysis (EDX).
3. Results and Discussion
Initial investigation of the diamond film growth surface by SEM (Fig.1(a)) showed that the (100) surface did not grow at a uniform rate over the whole substrate area. Differential growth rates in different areas of the surface created various levels of terraces separated by vertical growth steps. In addition, isolated crystals formed via secondary nucleation, showing that the deposition conditions were not perfectly optimised for homoepitaxy. Both these features were not unexpected, however, an unusual feature of the film surface was the presence of growth features with a ‘whisker-like’ morphology. These whiskers were mostly straight and appeared to taper to a point, with the one end located at the growth step edges on the diamond film surface (see Fig.1(b)). The whiskers had a width of <1 μm with an average length of ~20 μm, although some were as long as 50 μm. Some of the whiskers were observed to have peeled away from the surface and begun to curl up lengthwise (as if tending to a tubular structure), resulting in an appearance reminiscent of a ‘celery-stick’, see Fig.1(c). EDX analysis showed these whiskers to be composed solely of carbon. After the whiskers had been removed from the surface by washing with ethanol, ‘shadows’ remained behind indicating where the whiskers had lain. These shadows suggest that the whiskers had grown and peeled away from the surface at some point during the deposition process. They had then lain on top of the surface in such a way as to mask areas of the surface from the plasma, thus preventing growth underneath them. An example of this shadowing is shown in Fig.1(d), where the ‘ghost’ of a whisker can be seen both on the surface and crossing a secondary-nucleated diamond crystallite.




Figure 1. SEM micrographs of
the ‘celery-stick’ whiskers on the surface of the diamond film after CVD. (a) A low magnification picture showing the
large number of whiskers, secondary nucleated diamond crystallites, and the
step edges. (b) A higher magnification
picture showing a single whisker originating from and running along a
step-edge. (c) Whiskers that have fully
peeled away from the surface and curled up lengthwise to form
celery-sticks. (d) The ‘shadow’ of a
whisker that had masked an area of the surface during growth.
Figure 2 shows bright field TEM micrographs of some of the smaller of the ‘celery-like’ whiskers. The wide variation in the size of such whiskers is illustrated in Figure 2(a), in which the larger structure appears to be tubular or semi-tubular (with darker contrast at edges than centre), whereas those at the top and bottom of the micrograph appear solid and cylindrical or flat. Further examples of such growth are presented in Figure 2(b). Here some of the whiskers are filled with material (labelled as ‘A’) with a different appearance to that comprising the walls. Selected area diffraction was carried out on this section of the whisker and identified the material to be crystalline, although it also showed that it was not graphite. Similar investigations of areas (labelled as ‘B’ and ‘C’) that did not contain embedded material showed the whiskers to have an amorphous structure.
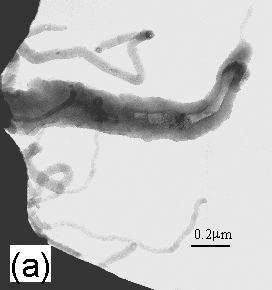

Figure 2. Bright field TEM
micrographs of some of the smaller of the whiskers. (a) A tubular or semi-tubular whisker (centre), with some solid
and cylindrical or flat whiskers (top and bottom). (b) Example of whiskers filled with crystalline material
(labelled ‘A’). The walls and bulk of
the whiskers (labelled as ‘B’ and ‘C’) are amorphous.
Due to their size, the larger celery structures seen previously in the SEM were difficult to analyse using TEM, but we believe that the smaller tube-like structures seen in Fig.2 may be ‘immature’ versions of the larger whiskers at an earlier stage in their growth. These smaller tubes are noticeably not as straight as the longer celery sticks, but this may be as a result of the TEM preparation method. It is also worth noting that the structures were stable under a 250 kV focused electron beam.
The small structures visible in Figures 2(a) and (b) have similar diameters (0.2-0.8 μm) to the carbon nanobeads reported previously [11], as discussed above. They also seem to have a bobbled structure reminiscent of a string of beads. The previous work also reports the presence of a metal catalyst (Fe) during the synthesis of the material, which has similarities to the present work in which the HPHT diamond substrate contained metallic impurities.
4. Conclusions
There are many similarities between the small tubular and larger celery structures presented here, and previously reported carbon nanobeads (Fe catalysed) and tubular growth. Along with the fact that the larger celery structures seem to be associated with growth step edges on the diamond surface, this seems to indicate that the growth reported here is due in some way to catalytic activity of metals present in the HTHP single crystal diamond substrate. Another important contributing factor may be the relative flatness of the growing diamond surface as this allows extended growth of the features observed.
We can therefore construct a growth sequence that is consistent with these observations, and this is illustrated in Fig.3. Uneven growth of the diamond surface during CVD produces vertical step-edges, which act as nucleation sites for fast lateral diamond growth. However, since the CVD conditions are not optimum for epitaxial growth, defects may form on the step as a result of secondary nucleation, leading to a distortion in the lattice at these points. Alternatively, metallic impurities within the HPHT substrate may diffuse to the surface due to the high deposition temperature, and act as a catalytic centre. In either case, subsequent growth that originates from the defect sites no longer follows the template of the diamond lattice. Instead, it results initially in many small tube-like amorphous carbonaceous growths, but eventually forms a single long, thin coating that grows along and coats the edge of the step. As the coating grows longer, stresses may develop which eventually cause it to partially peel away from the step edge. These same internal stresses may then cause the whisker to curl up lengthwise into celery-stick structures.
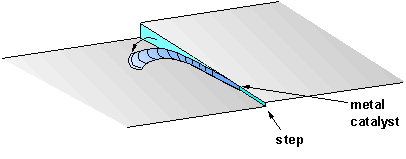
Figure 3. An illustration of the suggested sequence for formation of
the celery-sticks. Growth along the
step-edge originates from either a catalytic metal impurity or a secondary
nucleation defect, and then proceeds forming a long, thin coating which covers
the step. This then peels away and
curls up lengthwise.
Further work, including more high resolution TEM and electron diffraction analysis, needs to be done to ascertain the exact composition of the celery-sticks.