

Considered to be the most influential chemist since the 18th century founder of chemistry, Lavoisier, Linus Pauling applied quantum physics to chemistry and his discoveries affected the work of all chemists to follow.
Pauling is considered the father of molecular biology, which provided the base for biotechnology.
In 1954 Pauling received the Nobel Prize in chemistry for his work on the Nature of the Chemical Bond.
Protein / polypeptide structure:
In the late1930s, Linus Pauling and Robert Corey began x-ray crystallographic studies of the precise structure of amino acids and peptides. Their aim was to obtain a set of standard bond distances and bond angles for these building blocks and then use this information to predict the conformation of proteins. One of their important findings was that the peptide unit is rigid and planar. The hydrogen of the substituted amino group is nearly always trans (i.e. opposite) to the oxygen of the carbonyl group.
The bond between the carbonyl carbon atom and the nitrogen atom of the peptide unit is not free to rotate because this link has partial double-bond character.
Thus, there is a large degree of rotational freedom about these bonds on either side of the rigid peptide unit.
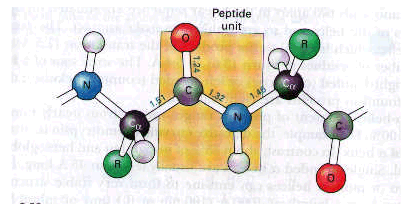
Can a polypeptide chain fold into a regularly repeating structure?
Pauling and Corey evaluated a variety of potential polypeptide conformations by building precise molecular models to answer this question. They adhered closely to the experimentally observed bond angles and distances for amino acids and small peptides. In 1951, they proposed 2 periodic polypeptide structures, called the alpha helix and the beta pleated sheet.