

|
|
|
|
|
|
|
|
|
Glossary of unfamiliar terms
adenosine triphosphate, ATP and lesser phosphorylated relations, ADP and AMP (di- and mono- respectively) are yet more examples of the importance of the chemical purine. As mentioned on the methyl xanthines page, purine is used to form adenine and guanine, two of the four basic constituents of the nucleotides that are used to construct DNA and RNA, found within every living organism and carrying the genetic code that is the basis of life. Also important is the addition of a ribose sugar to adenine and guanine to make adenosine and guanosine respectively. The phosphorylated forms can be hydrolysed, e.g. ATP going to ADP and inorganic phosphate, yielding 30.6kJ/mol of energy. All known living cells use this reaction when they need energy to perform work. Thus ATP is known as the 'universal energy carrier' or 'energy currency'. The use of reactive ATP in cellular reactions as a template is another example of the significance of ATP in biochemistry.
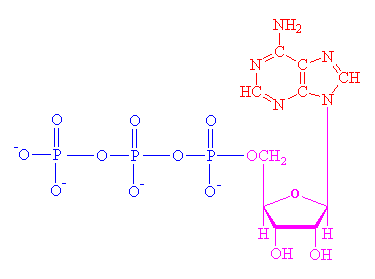
Further reading: Paul May's site www.bris.ac.uk/Depts/Chemistry/MOTM/atp/atp1.htm
alkaloid organic compound found
in plants whose name derives from its basicity - in ancient times these
compounds were extracted from plants using aqueous acid and then precipitation
by neutralization.
neuromodulator small, diffusible molecules
that control nerve impulses passing through a region called the synaptic
cleft. The presynaptic membrane of a synapse is separated from the postsynaptic
membrane by a gap of about 50nm, and chemicals have to cross this gap to
communicate the nerve impulse.
and Biological Science 1,
Taylor, Green
and Stout, Cambridge Press, 3rd Ed. 1997
Further reading: Organic Chemistry,
Clayden, Greeves, Warren and Wothers, OUP, 2001
Further reading: Biochemistry, Stryer,
Freeman Press, 4th Ed. 1995