Production
Technetium has never been found in a naturally occurring terrestrial material. It has however been detected in the spectrum of various stars, the discovery of which is currently influencing theories of production of other heavy metals in stars.
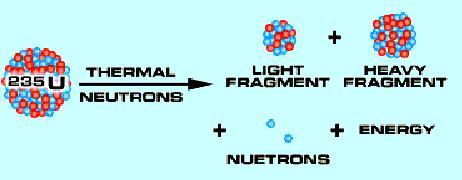
Technetium can be manufactured in a couple of ways. The most common is by the bombardment of molybdenum with deuterium nuclei. Molybdenum is a by-product produced in nuclear reactors and is produced by up to 6% of all uranium-235 thermal fissions.

The Molybdenum is then purified and then converted to Technetium. This process generates the most stable isotope, Technetium-98. However this isotope is not the most important to nuclear medicine. That honour is reserved for 99mTc.
99mTc is obtained from the radioactive decay of molybdenum-99 in a molybdenum-technetium generator, in the form of sodium pertechnetate (Na99mTcO4). 99mTc decays by isomeric transition to 99Tc and it has a half-life of 6.02 hours. It is the radioactive decay that emits g radiation which is used in nuclear medicine.