Chemistry of Coral Skeleton and Mollusc Shells
The tiny coral polyps secrete a limey substance that hardens into the coral structure. The substance is calcium carbonate (CaCO3) with a small amount of specialized proteins also present in the coral skeleton.
There are two forms of calcium carbonate, aragonite and calcite. Their crystal shape differs though their chemical formula is the same. Aragonite is the denser form and is found in the hard or scleractinian corals. Whether the aragonite or calcite form is produced depends on the “seed crystals” growth pattern and the proteins secreted by the polyps.
The aragonite form is also found in mollusc abalone shells. These shells have a “mother of pearl” layer in which this nacre form of calcium carbonate was thought to be produced in part, by specialised proteins found in the mollusc shells. The proteins were separated, isolated and identified by polyacrylamide gel electrophoresis, electro-elution, and amino acid analysis.
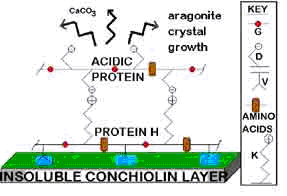
The proteins of shells are generally acidic though a basic protein has also been found. It is thought to bind the insoluble matrix to acidic proteins in such a way that promotes the mother-of-pearl formation. The diagram shows how this complex of matrix, basic protein H, and acidic proteins are orientated in such a way that the mother-of-pearl is formed. The amino acids Glycine (G), aspartic acid (D), valine (V), and lysine (K) are present in relatively high amounts in protein H.
Research has been done on comparing the similarities of proteins in molluscs and the coral skeleton as they both have the aragonite form of calcium carbonate. The amount of protein found in the abalone shell was much greater than in the coral. The amino acid content was dissimilar between the shell and coral, indicating a different role or function in the formation of the calcium carbonate skeleton. Also showing that because the protein H is found in the nacreous aragonite but not in non-nacreous aragonite, this protein’s presence may be the necessary factor in the formation of the mother-of-pearl.