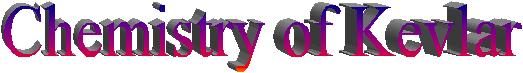The basic unit
Kevlar is a polymer; this means that it is made
up of a large number of the same basic unit, called a monomer, which are
attached to each other to form a long chain.
The monomer in this case is made up of an amide group and a phenyl
group.
Kevlar is a polyamide, a type of synthetic
polymer, in which the amide groups are separated by para phenylene groups,
meaning that the amide groups are attached to each other on opposite sides of
the phenyl group (i.e. carbons 1 and 4).
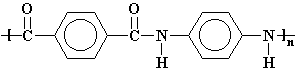
The large
phenyl groups separating the amides cause the polymer of Kevlar to nearly
always form the trans conformation, where the phenyl groups arrange themselves so
that they are on opposite sides of the rigid amide bond:
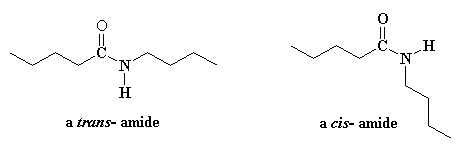
This is
caused by the phenyl groups to be too large to fit on the same side of the
bond, as there would be great steric hindrance between the hydrogen atoms:
The hydrogens are too close together in the cis conformation In the trans conformation there is much more room, and
so less hindrance.
![]()

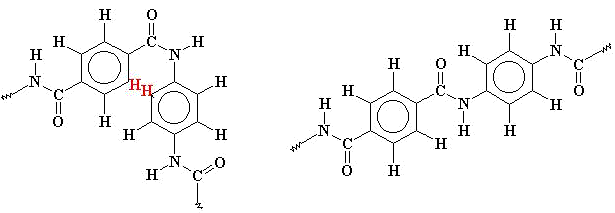
This
situation where all the monomers connect in a trans conformation, means that
very long straight chains are formed, making an almost ideal fibre, and allowing it such a wide range
of uses.
More information on why Kevlar forms such
straight long Chains