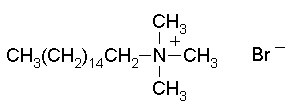
The main components of shampoo and conditioner are surfactants. These are molecules made up of a hydrophilic head group and a hydrophobic carbon chain.
An example of a surfactant commonly used in shampoo is sodium laureth sulphate. This is an anionic surfactant, meaning that it has a negatively charged head group. Its function in shampoo is to provide a lather.
Non-ionic surfactants are not often used in shampoos as they can strip the hair and cause scalp irritation. Very mild ones are used as foam stabilisers and thickeners, and they are used in shampoos for greasy hair.
Amphorteric surfactants are used frequently because they decrease irritation whilst increasing the active contents level and the quality of the lather produced. The are so mild that they are often used in baby shampoos.
The main component of conditioner is a cationic surfactant such as cetyl alcohol. This sticks to the surface of the hair by static attraction, because the hair carries a slight negative charge when wet, so the positively charged head group of the surfactant is attracted to it. The conditioner is therefore difficult to remove from the hair, so it smoothes the hair and coats it, making it easier to comb.
A common conditioner ingredient is cetrimonium bromide, shown below. This is an anti-static agent so improves the look and shine of the hair. Glycerol, mineral oils or a silicone polymer are often added to less expensive conditioners to add shine to the hair. These can accumulate if used constantly in 2 in 1 formulations, leading to lank and greasy hair.
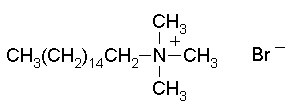
More expensive conditioners contain complex ammonium polymers. These have a more subtle effect on the hair and are less likely to lead to lankness due to build up. Hydrolysed silk and other hydrolysed proteins give shine and flow, while keratin amino acid helps improve the strength of the hair by adding keratin from which the hair is made.
|
|
Colouring |
|
|
Parfum - only listed as parfum on packaging as the ingredients are too complicated to list fully |
|
|
Preservatives - these kill a wide range of microbes which are likely to spoil the product |
|
|
Sodium chloride - this provides a cheap way of thickening the product |
|
|
Citric acid - rebalances the pH of the product, which is slightly alkaline by nature |
|
|
Tetrasodium EDTA - a water softening agent. Hard water can be a problem if calcium and magnesium salts have not been removed, as the counterion in the anionic surfactants can be replaced by Ca2+ or Mg2+, which make the surfactant less soluble and results in a cloudy product. |