Contact lenses are becoming increasingly popular with wearers of glasses as their comfort and ease of use increases. Maximum contact lens comfort is achieved when the eye can 'breathe', so they are made from gas - permeable material. Silicone is the material of choice because it can be modified in many ways to suit different specifications. Silicones are also inert, so do not cause reactions in the eye. This is why they are also used in the body, for example as breast implants.
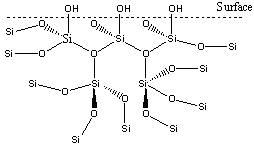
Silicones are primarily made up of silicon and oxygen. They are cross-linked polymers with a functionalised surface.

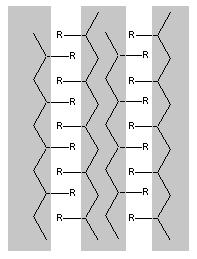
If the OH groups on the surface are replaced with an alkyl chain, represented by R, the surface becomes hydrophobic. If this is long or bulky enough the structure changes, creating hydrophobic channels in the solid. This makes the material gas - permeable and a good oxygen carrier, thus preventing the eye from drying out.
The surface of the lens in contact with the eye must be hydrophilic so that the lens is not repelled from the wet surface of the eye. This presented a problem in the earliest contact lenses as the first silicone lens materials were naturally hydrophobic, leading to discomfort and often damaging the cornea. Water permeated through the lenses from the surface of the eye, but was unable to return to the eye tissue under the lens, resulting in dehydration of the tissue, as well as adhesion of the lens to the eye. This problem was resolved by grafting hydrophilic polymer systems onto the silicone polymers.