Their capability of inducing muscle paralyses lends Botulinum Toxins to the treatment of several medical conditions, such as Cervical Dystonia, involving spasms of the neck muscles causing a tilting of the head. A low strength form of Botulinum Toxin type A (or trade name Botox®) can be administered to relieve this problem. When the toxin is injected into a specific area it results in paralysis of the local muscle only, leaving the surrounding muscles able to function properly. This is the principal behind all known medicinal and cosmetic treatments using Botox®.
Some side effects of such treatment are most commonly reported to be neck pains and headaches. However such effects did not stop Botox® from receiving official approval for the treatment of Cervical Dystonia in the year 2000.

Above : A case of Cervical Dystonia the main symptom is involuntary movement of the neck.
Source - Modified from http://www.dystoniafoundation.org
Botox® has also been used to relieve the condition of excessive blinking of the eyelids, (Blepharospasm) by injection directly to the affected eyelid. However the resulting side effects are found to most commonly be corneal ulceration due to over exposure (i.e. due patient not blinking enough!), skin rashes and swelling of the eyelids. Similar problems have been reported when treating misalignment of the eyes (Strabismus) with Botox®.
Strabismus and Blepharospasm were amongst the first conditions to be treated with Botox®, with official approval being granted in 1989.
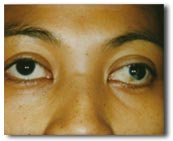
Above : A case of Strabismus or crossed eyes, conventionally cured by operation, can also be cured by a localized Botox® injection.
In May of 2002 scientists revealed their studies regarding the successful use of Botox® in treating bladder problems. The study claimed that 82% of the people with incontinence surveyed, reported a dramatic improvement in bladder control in the six months following treatment with Botox®.
Bladder problems joined Juvenile Cerable Palsy, amongst other clinical conditions, in the early stages of development in using Botulinum Toxin type A for their treatment, and have yet to gain official approval for their implementation.