Chemistry 1S Dr. Paul May, S103A, S114
1. What is Calculus?
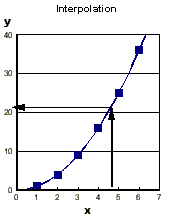
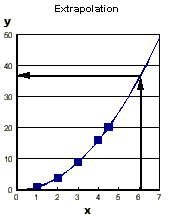
In physical chemistry we try to represent measured quantities
of some variable (say, temperature or pressure) by a function.
This allows us to interpolate between the measured points
to find a value for a point not measured, and extrapolate
to find a value for a point outside the range.


Examples of functions
1. How is the pressure of a gas related to other measurable properties,
such as volume and temperature?
Answer: The Ideal Gas Eqn. p = 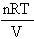
The pressure, p, is said to be a function of 3 variables:
i) n, the number of moles of gas
ii) T, the temperature of the gas
iii) V, the volume of the gas.
R is a constant ( = 8.314 J K-1 mol-1)
Now, chemistry is the science of things changing, so as
physical chemists we are interested in finding out how and why
things change, and how fast, and if we can measure it.
Changes are often observed as either slopes of graphs,
or areas under graphs.
slopes = differentiation, area = integration
Example of a slope
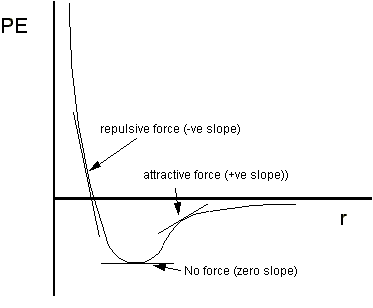
2. How does the force between two molecules depend upon their
potential energy, PE, and their separation, r ?
Answer: From physical chemistry lecture notes, you'll learn that
the force is given by the slope of the PE
vs r graph at any point.
Example of an Area
3. How much heat energy must we provide to raise the temperature
of a liquid from 0C to 100C, given that its heat capacity is a
function of temperature?
Answer: The amount of heat energy needed is given by the area
under the heat capacity vs. T graph.

2. Functions and Graphs
If the value of a quantity, A, is dependent upon the value
of another quantity, B, then we say A is a function
of B. (Note also, that B is obviously a function A
as well.)
Examples:
Rather than write out ' ... is a function of ...' each time, we
use an abbreviation of the form f(x), which is read
as 'f of x'. We replace the f by the dependent
quantity, and the x by the variable name.
Examples
e) From (c) above, if rate is R and temperature is T, then the shorthand for (c) is to write R(T).
f) If energy is E and frequency is ν, then the shorthand for (d)
is E(ν).
This means that if we know the value of the symbol in brackets,
we can calculate the value of the symbol outside the brackets.
- if we know the frequency of a photon, we can work out its energy.
For most examples we'll use the y(x) notation, where
we know the value of x and want to calculate y,
e.g.:
y(x) = x2
y(x) = 2x + 3 , etc.
but you should realise that in chemistry we are dealing with quantities
in the real world, and so we 'try' to give them meaningful symbols
(p for pressure, V for volume, etc.). Therefore we often
get functions like
A(r) = πr2 (Area of a circle),
F(C) = 32 + 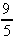 C (Fahrenheit to Centigrade),
C (Fahrenheit to Centigrade),
p(V) = 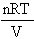 (Ideal Gas Eqn., at constant
n and T),
(Ideal Gas Eqn., at constant
n and T),
or F(r) = 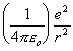 (Inverse square law
- Coulomb force between 2 electrons)
(Inverse square law
- Coulomb force between 2 electrons)
Sometimes we use Greek letters for variables, e.g.:
E(ν) = hν (E = energy, ν= frequency, h = Planck's constant)
λ(ν) = c / λ (ν= frequency, λ= wavelength, c = speed of light)
or even new, 'invented' symbols, e.g.:
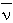 (&lambda) = 100 / &lambda (
(&lambda) = 100 / &lambda (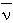 = wavenumber in cm-1, &lambda = wavelength in m)
= wavenumber in cm-1, &lambda = wavelength in m)
[A](t) = [A]o e-kt ([A] = concentration
of chemical A, t =time, k= rate constant, e = exponential
function, see later)
Sometimes a function can depend upon more than one variable.
In this case we write all the variables in the bracket. For example,
if y is a function of x and t, we write y(x,t).
Examples
i) The free energy, G, of a reaction depends upon the enthalpy, H, and the entropy, S.
So,
G(H,S) = H - TS (T is constant, H and S vary)
ii) If n and T can vary too, the Ideal Gas Eqn. given earlier
becomes a function of 3 variables, and is written
p(n,V,T) = 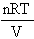
Mostly during this course, we'll restrict ourselves to dealing
only with functions of one variable.
 Next lecture
Next lecture