
Smog in Korea
[Image: DestinationFearFan, CC BY-SA 4.0 via Wikimedia Commons] |

Peroxyacetyl nitrate (PAN)
(a.k.a. Acetyl nitroperoxide)
The eye-irritant in smog

Simon Cotton
University of Birmingham

Molecule of the Month March 2026
Also available: HTML version.

|
|
That is a scary looking molecule!
Indeed, and it does scary things.
How so?
It’s irritating, like you.
What’s it like?
It is a very unstable molecule, explosive in bulk. And it’s like being kicked in the goolies.
How so?
It makes you cry.
How does that happen?
Peroxyacetylnitrate is found in city air, especially on a hot day, when there is lots of traffic around. It is an eye irritant, something to do with that peroxo group; its instability; as well as the likelihood of its forming acidic stuff like NO2. Your eyes recognise it as a ‘foreign’ molecule, and your tear ducts produce fluid (tears) to wash it away. It also can irritate the respiratory system.
|

Smog irritates your eyes causing tears
[Image: Mimooh, CC BY-SA 3.0 via Wikimedia Commons] |
And how can it be made?
A mixture of diacetyl, nitrogen dioxide and oxygen was irradiated at around 0 °C using a mercury arc-lamp as the light source for the photolysis.
CH3C(O)C(O)CH3 + NO2 + O2  CH3C(O)OONO2
CH3C(O)OONO2
After irradiation, the mixture was distilled through a trap at -80 °C. A mixture of PAN and unreacted diacetyl, plus some methyl nitrate, was obtained from the trap and separated by glc (gas-liquid chromatography).

Panorama of the smog in LA in 2005.
[Image: Photo by DAVID ILIFF. License: https://creativecommons.org/licenses/by-sa/3.0/]
How was it discovered in the first place?
It is the simplest member of the series of peroxyacyl nitrates (PANs), discovered on 1956.
Its discovery is to do with SMOG. Although the word smog (SMoke + fOG) was chosen to describe the air pollution of Los Angeles, it contained neither smoke nor fog. Indeed, the smog in LA became so notorious in the 1940s and 50s that the author Raymond Chandler featured it in many of his Philip Marlowe detective novels, using the smog as a metaphor for the corruption, and moral decay that permeated the seedy underworld featured in his novels.
"I smelled Los Angeles before I got to it. It smelled stale and old like a living room that had been closed too long.", Raymond Chandler, The Little Sister, 1949.
Who worked out the cause of smog?
Haagen-Smit.
You mean, the ice-cream man?
No, that is Häagen-Dazs. The Dutch-born Arie Jan Haagen-Smit (1900-1977) was an organic chemist - he did his PhD for the great Leopold Ruzicka - who settled in the USA as an academic at Caltech.
Haagen-Smit determined that the Los Angeles smog arose through the action of sunlight in the presence of automotive vapours. His reasoning was that LA smelt like an organic chemistry laboratory, perhaps a place where ozonolysis was being carried out.
|

Arie Jan Haagen-Smit (1900-1977)
[Image: Interest Group - Environmental Chemistry] |
 So where did the ozone originate?
So where did the ozone originate?
He deduced that it had to be produced in the atmosphere, so that pollutants arose from sunlight acting upon organic compounds from automobile exhausts.
How is PAN formed in polluted air?
Incompletely combusted fuel in internal combustion engines leads to radicals like CH3CO. This reacts with atmospheric oxygen generating CH3CO-OO.
CH3CO + O2  CH3C(O)O2
CH3C(O)O2
This, in turn, goes on to attack the traces of nitrogen dioxide (MOTM December 2012) forming CH3CO-OONO2; here M represents a ‘third body’ such as a N2 molecule.
CH3C(O)OO + NO2 + M 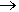 CH3C(O)OONO2 + M
CH3C(O)OONO2 + M
 Where does the nitrogen dioxide come from?
Where does the nitrogen dioxide come from?
CH3CO-OO reacts with nitric oxide, NO, oxidising it to NO2. This is quite important during morning smogs. By the afternoon, the NO has been used up, so the reaction between CH3CO-OO and NO2 becomes important.
Why is PAN so toxic?
The PAN molecule contains so much oxygen, so it oxidises organic matter – including plant leaves.
Can anything be done about this?
Many city councils around the world are introducing 'clean air zones' where only more modern cars that burn petrol more efficiently producing less pollutants and particulates are allowed in freely - older, more polluting vehicles need to pay a hefty charge. And, of course, the transition to electric vehicles may eventually remove smog from our cities completely, but only if the electricity is also generated in a clean way, such as via solar, wind or nuclear power.

Bibliography
- CRC Reference Number: DNF55-K
- A.J. Haagen-Smit, Ind. Eng. Chem., 44 (1952) 1342-1346 (pioneering study of Los Angeles smog)
- A.J. Haagen-Smit, C. E. Bradley and M. M. Fox, Ind. Eng. Chem., 45, (1953) 2086-2089 (Ozone formation and the photochemical oxidation of organic substances)
- A.J. Haagen-Smit, Sci. Amer., 210, (1964) 25-31 (‘The control of air pollution’)
- E.R. Stephens, F.R. Burleson, E.A. Cardiff, J. Air Pollut. Control Assoc., 15, (1965) 87- 89 (synth)
- J.E. Lovelock and S.A. Penkett, Nature, 249, (1974) 434 (‘PAN over the Atlantic and the smell of clean linen’)
- A.J. Kacmarek, I. J. Soloman, M.Lustig, J. Inorg. Nucl. Chem., 40, (1978) 574- 575 (synth, props)
- P.B. Shepson, T.E. Kleindienst, E.O. Edney, C.M. Nero, L.T. Cupitt, L.D. Claxton, Environ. Sci. Technol., 20, (1986) 1008-1013 (toxicity)
- L.F. Joos, W.F. Landolt, H. Leuenberger, Environ. Sci. Technol., 20, (1986) 1269-1273 (properties)
- J. M. Roberts, Atmos. Environ. 24A, (1990) 243-287 (review of the atmospheric chemistry of organic nitrates)
- A. Vyskocil, C. Viau, S. Lamy, Human & Exper. Toxicol. 17, (1998) 212-220 (Peroxyacetyl nitrate toxicity reviewed)
- T.A. Teklemariam J.P. Sparks, Plant, Cell and Environment, 27, (2004) 1149–1158 (PAN toxicity to plant leaves)
- A.M. Holloway R.P. Wayne, Atmos. Chem., 2010, Cambridge, RSC Publishing, esp. pp. 106-107, 216-219 (PAN and SMOG)
- A. Zare, P.S. Romer, T. Nguyen, F.N. Keutsch, K. Skog, R.C. Cohen, Atmos. Chem. Phys., 18, (2018) 15419–15436. (the lifetime of atmospheric organic nitrates)
- L.T. Molina, Faraday Discuss., 226, (2021) 9–52 (air quality in megacities)
- P. Brimblecombe, Arie Jan Haagen-Smit and the history of smog.


 Back to Molecule of the Month page. [DOI:10.6084/m9.figshare.29468423]
Back to Molecule of the Month page. [DOI:10.6084/m9.figshare.29468423]

![]()
![]()
![]()
![]()


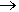 CH3C(O)OONO2
CH3C(O)OONO2

 So where did the ozone originate?
So where did the ozone originate?  Where does the nitrogen dioxide come from?
Where does the nitrogen dioxide come from?