![]()
Para-Phenylenediamine (PPD)
The hair dye that causes skin irritation
![]()
![]()
Molecule of the Month October 2022
Also available: HTML version.
![]()

Para-Phenylenediamine (PPD)The hair dye that causes skin irritation
Molecule of the Month October 2022
|
 |
No, the exact opposite. Hydrogen peroxide (MOTM sept 2006) bleaches hair and makes it lighter. PPD makes hair darker.
When applied to hair, PPD oxidises and turns hair into a dark colour which doesn't fade over time. It is therefore used as the main ingredient in permanent hair-dye formulations. In the USA, one third of all women use hair dyes containing PPD and it is found in over 1000 hair dyes around the word.
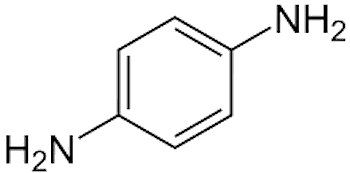 |
|
| Para-phenylenediamine, a.k.a. PPD, 1,4-diaminobenzene |
It’s also used to dye fur and to darken cosmetics, as well as to intensify the black colour produced by henna in temporary tattoos.
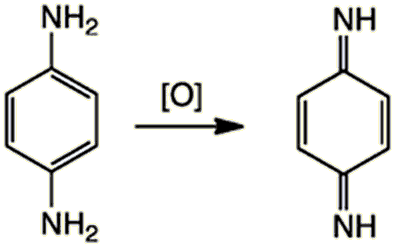
The first step involves oxidation of PPD (usually using hydrogen peroxide) to its quinone form under basic conditions (e.g. in ammonia):
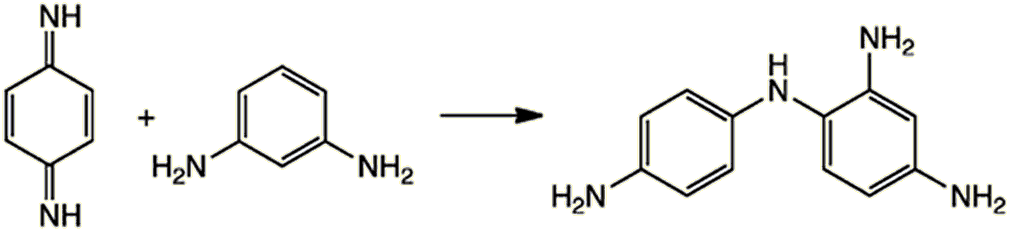
Then an electrophilic aromatic substitution occurs with a coupler molecule, such as 1,3-diaminobenzene. The type of coupler chosen determines the colour of the final dye.
In the third and final step, the product from this reaction oxidiszes to the final hair dye.
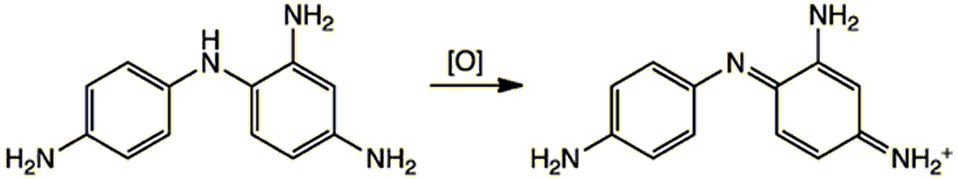
These larger dye molecules get locked inside the hair, permanently colouring it.
But what is PPD?PPD is a crystalline solid which can have a variety of colours including pink, grey or yellow or even colourless. On exposure to air it oxidises and turns red, brown, then finally black. It was first synthesised by A.W. Hofmann in 1863, who was actually the chemistry professor of William Henry Perkin who made the world's first synthetic dye mauvine (MOTM January 1996). Nowadays, nearly all PPD is produced synthetically, but it can also be found as a black mineral from the banks of River Nile. In Africa, the Middle East, and India, PPD is commonly mixed with henna (Lawsonia inermis), which is traditionally applied to decorate the palms of hands, soles and feet and to dye hair a dark red shade, frequently for special occasions such as weddings. |
 A temporary henna tattoo. [Photo: AKS.9955, CC BY-SA 4.0 via Wikimedia Commons] |
You guessed it! PPD has been reported to cause mild reactions such as skin irritation and rashes. But there are also reports of people getting more serious conditions such as skin inflammation and eczema, and even severe dermatitis with pronounced reddening and swelling on the face and scalp. In fact, PPD was declared “Contact Allergen of the Year” in 2006 by the American Contact Dermatitis Society!
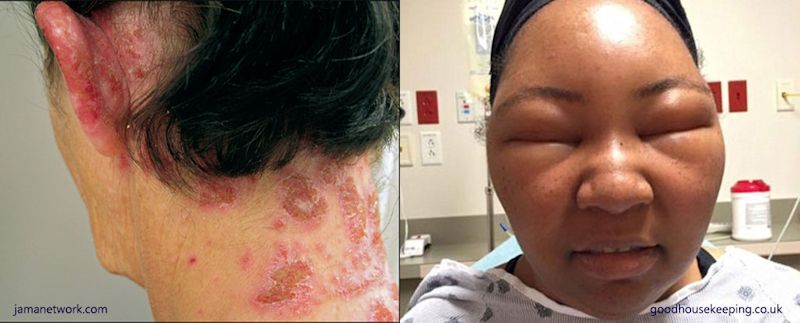
Allergic reactions to PPD, including skin rashes, demititis and swelling of the face and scalp.
 I think I remember the doctor from NCIS having this!
I think I remember the doctor from NCIS having this!Yes, that's correct and it made quite a sensational news story in 2014. Pauley Perrette (photo, right) is the actress who played the gothic forensic scientist Abby Sciuto, noted for her very black dyed hair, in the US TV series NCIS. One day after dyeing her hair, she had an extreme allergic reaction - her face swelled up and she had to go to the Emergency Clinic. She tweeted photos of her swollen face as a warning to others about the dangers of 'hair dye allergy'.
The most frequent serious health consequence from PPD is sensitisation to hair dye and related chemicals. This can come from simply using hair dye containing PPD, but often arises from people who get a ‘black-henna’ temporary tattoo. ‘Black henna’ is a misleading name because it is not derived from the henna plant, but instead from indigo (MOTM Feb 2009), and is often a mixture of other unlisted dyes and chemicals, plus PPD. If a person previously had a ‘black-henna’ tattoo (or a tattoo made from normal henna mixed with PPD) and had an allergic reaction, they may have become sensitised to PPD. If they subsequently dye their hair with a PPD-containing hair-dye, a far more serious allergic reaction may occur which can be life threatening and require hospitalisation. In recent years there has been an ‘epidemic’ of PPD allergic reactions – so much so that chemical hair-dye products now post warnings on the label. 'Hair-dye allergy' can go much further - once sensitised, people can also have allergic reactions to molecules similar to PPD, such as para-aminobenzoic acid (found in sunscreens products), sulfonamides and sulfonylureas (in medications), azo dyes (used in clothes, ball point pen ink, and shoes), benzocaine, tetracaine, procaine (in local anesthetics), and para-amino salicylic acid (in antibiotics). So, these unfortunate people are suddenly allergic to almost everything around them!
Animal tests have shown that PPD can also rapidly pass through the skin and cause a range of systemic effects including tremors, drowsiness, convulsions, liver atrophy, cardiac arrest, and dark urine – which becomes a characteristic chocolate brown colour!
This probably isn’t a problem with the low exposure a person receives when dyeing their hair, especially as the amount of PPD in hair products is restricted to 2-6% (depending on country), and application instructions state that that the dye must not touch the scalp and must be quickly rinsed away. (In contrast, the illegal ‘black henna’ pastes may contain 10% to 80% PPD and are left on the skin for 30 minutes!). However, for hairdressers who use these dyes regularly and often don’t wear gloves, there’s a potential risk.
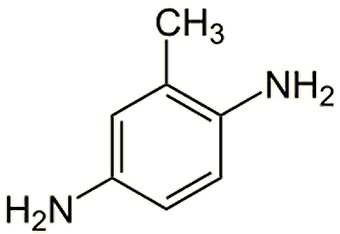
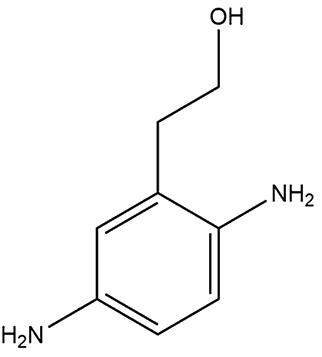
Not surprisingly, alternative, safer dyes are being developed. The include 2,5-diamino(hydroxyethyl)benzene and 2,5-diaminotoluene which are both less toxic than PPD. Others include derivatives of tetraaminopyrimidine, indoanilines and indophenols.
 |
 |
||
| 2,5-diaminotoluene | 2,5-diamino(hydroxyethyl)benzene |
| A group in Singapore (see Venkatesan et al., below) is also studying molecules derived from PPD where the ortho-group R is modified via the addition of hydrophilic ether functional groups to potentially make dye compounds that are more water soluble, and also less reactive towards the proteins found in skin. They tested seven compounds which all worked well as hair dyes, with many of them exhibiting a reduced inflammatory response. New derivatives such as these may one day replace PPD in all hair dyes. | 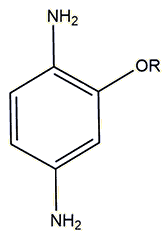 The R group is modified to a range of short-chained ether groups, such as -CH2CH2OCH3 |
 Can PPD be used for anything else?
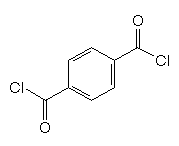
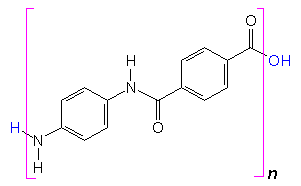
Can PPD be used for anything else?Yes, because PPD has 2 functional groups, it is very useful in polymerisation reactions. It is the basis of Twaron, a heat resistant polymer fibre, and Kevlar (MOTM Nov 2010), the strong fibre that’s used in bulletproof vests.
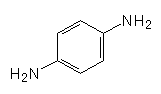 +
+ 
![]()
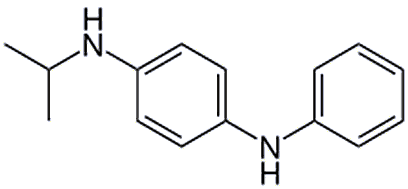
 Also, because it oxidises easily, a derivative of PPD called IPPD is used as a rubber additive to prevent damage to the rubber from ozone. The IPPD reacts with ozone faster than ozone reacts with the surrounding rubber. However, IPPD is also a human allergen, and is responsible for coining the term "Volkswagen Dermatitis". This condition was named after a patient in 1969 who washed his Volkswagen’s car tyres (which contained IPPD at the time) by hand and contracted dermatitis.
Also, because it oxidises easily, a derivative of PPD called IPPD is used as a rubber additive to prevent damage to the rubber from ozone. The IPPD reacts with ozone faster than ozone reacts with the surrounding rubber. However, IPPD is also a human allergen, and is responsible for coining the term "Volkswagen Dermatitis". This condition was named after a patient in 1969 who washed his Volkswagen’s car tyres (which contained IPPD at the time) by hand and contracted dermatitis.
 |
|
| N-Isopropyl-N'-phenyl-1,4-phenylenediamine, IPPD |
![]()
![]()
![]() Back to Molecule of the Month page. [DOI:10.6084/m9.figshare.20394342]
Back to Molecule of the Month page. [DOI:10.6084/m9.figshare.20394342]