
Cyclo-octasulfur, S8
Known as ‘brimstone’ in ancient times

Simon Zhang and Stephen Belding
Rugby School, UK

Molecule of the Month February 2026
Also available: HTML version.

|

Sulfur gathered from a volcano in Italy
[Image: Jan Helebrant, CC BY-SA 2.0 via Wikimedia Commons] |
“But the fearful, and unbelieving, and the abominable, and murderers,…
and all liars, shall have their part in the lake which burneth with fire and brimstone” - Revelation 21.8.
Cyclo-octasulfur is just the chemically accurate name for ordinary sulfur, right?
Correct, cyclo-octasulfur is the name given to the most abundant isomer of sulfur, the one we are all familiar with as yellow rocks or powder.
Why was sulfur historically known as brimstone?
Since antiquity, yellow deposits of sulfur were found close to active volcanoes, alongside molten rocks, fiery lava, and noxious gases (such as hydrogen sulfide, H2S). As a result, it was believed to be a substance that originated in Hell. In fact, sulfur is mentioned 15 times in the Bible. The term ‘fire and brimstone’ was often used in the Bible as terrifying description of the consequences of defying God, and the term is still used nowadays for a vociferous style of Christian preaching that promises that eternal damnation will befall sinners unless they repent.

Sulfur miners in the volcano Kawah Ijen, Indonesia, surrounded by toxic fumes
of H2S and SO2 (and water vapour). Yellow elemental S can be seen
in the collection baskets and on the ground.
[Image: Jean-Marie Hullot, CC BY-SA 4.0, via Wikimedia Commons]
‘Brimstone’ actually means ‘burning stone’, and the ancient Greeks and Romans burnt it as a fumigant to kill pests. It was also used as a bleach for clothes, disinfectant for surfaces, and for wine preservation. In 9th-century China, it was a key component to make gunpowder.
Is it that deadly?
Pure, elemental sulfur is non-toxic and is safe to touch. The picture (right) shows the common sulfur powder you might see in a school laboratory. However, many compounds of sulfur are toxic. For example, the gas hydrogen sulfide (H2S - the smell of rotten eggs, MOTM for March 2009) can inhibit cellular respiration and cause respiratory paralysis. Inhalation of sulfur dioxide, SO2 (MOTM for December 2003) at concentrations above 100 ppm can be fatal; even at lower levels, prolonged exposure can cause breathing difficulties, nausea, and corrosion of the airways. Carbon disulfide (CS2) can be irritating to your skin and cause dizziness. Furthermore, mustard gas (bis(2-chloroethyl)sulfide), is a powerful chemical weapon that was used in WWI (see MOTM June 1998).
|

Jar of sulfur powder
[Image: Self taken] |
Why is sulfur sometimes spelled with a ‘ph’?

Webster’s American Dictionary of the English Language 1828
[Image: Cullen328, CC BY-SA 3.0 via Wikimedia Commons] |
The original word came from the Latin word, sulpur, which had morphed into sulfur by the 3rd century. Even though it was derived from Latin, English scholars then applied the rules of the Greek language to this, which spelled the ‘f’ sound of ‘phi’ φ with a ph. We still have this legacy in many common modern Greek-derived words such as photograph, phlegm and phosphorus. Thus, the spelling became sulphur, which became the standard for centuries.
But when American lexicographer Noah Webster published his Webster’s dictionary of ‘American English’ in 1828, he simplified many of the spellings away from those used in Great Britain. For example, colour became color, centre became center, realise became realize, etc, and sulfur reverted to its Latin spelling. For nearly 200 years there were 2 different spellings for sulfur/sulphur, depending upon which side of the Atlantic you were on.
In 1990, the International Union of Pure and Applied Chemistry (IUPAC) standardised that sulfur is the spelling in all scientific documents (and likewise, sulfite, sulfate, sulfuric acid, etc). Nevertheless, 'sulphur' still remains in common usage in general British English outside of the scientific arena.
|
What shape is elemental sulfur?
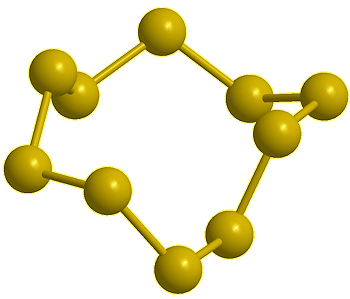
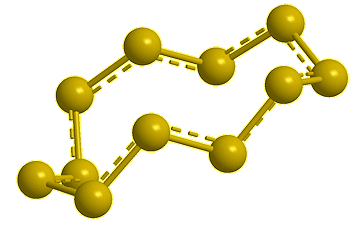
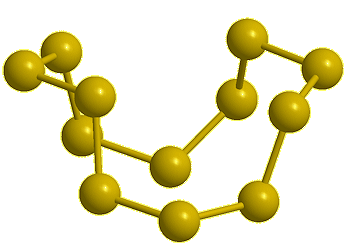
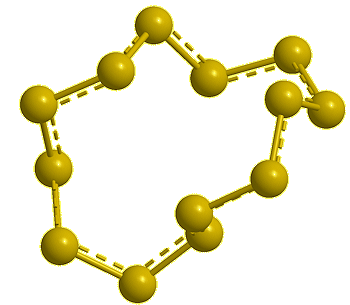
If you mean its molecular shape, then its most common isotope is a ring of eight S atoms with bond angles of 107.8°, and its scientific name is cyclo-octasulfur (S8).
It looks like a crown...
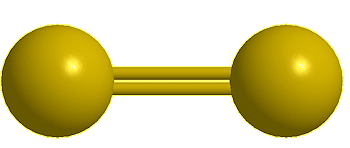
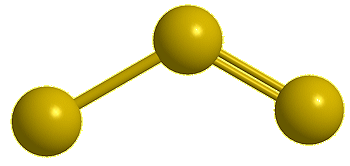
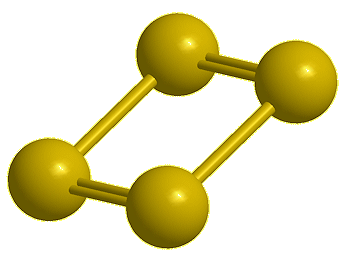
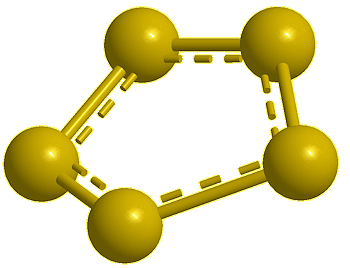
Yes, the common S8 molecule does look like a crown, but sulfur has other allotropes that form under different conditions, and sulfur molecules containing 2, 6, 7, 9–15, 18, and 20 atoms are known.
|
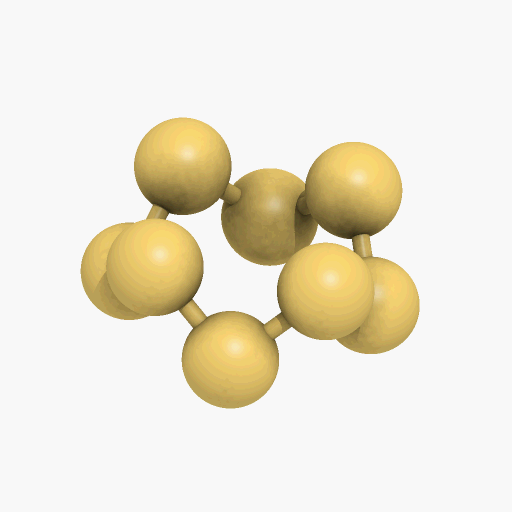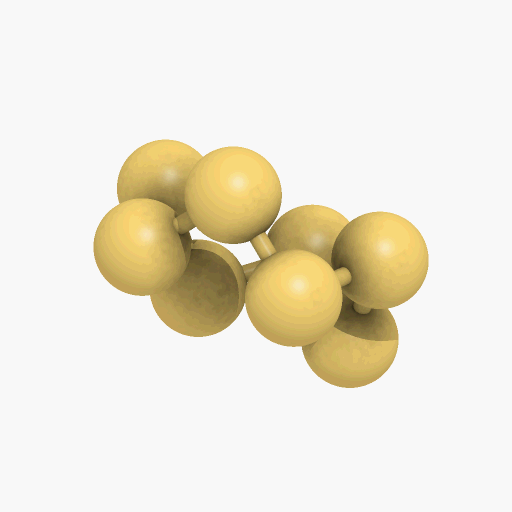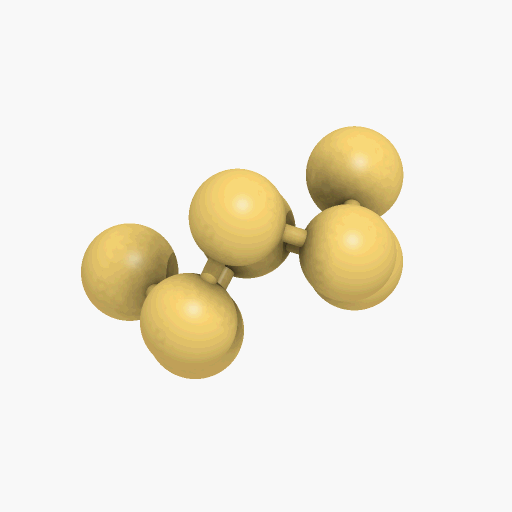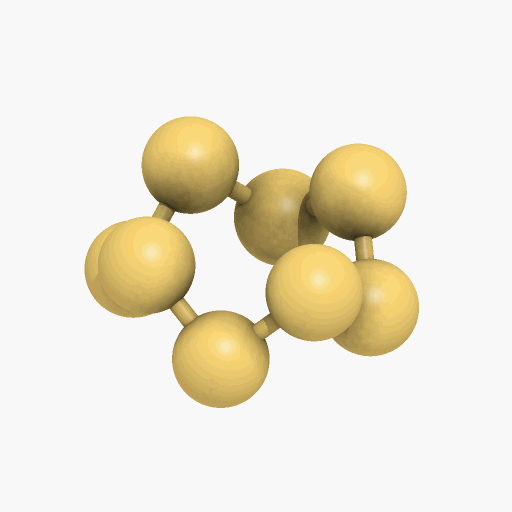
Structure of the S8 molecule
[Image: Manuel Almagro Rivas, CC BY-SA 4.0, via Wikimedia Commons]
|
 |
 |
 |
 |
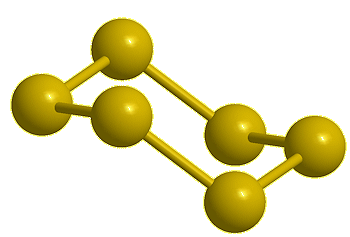
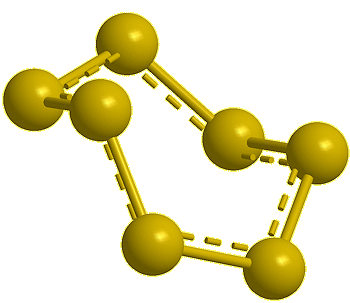
| Disulfur, S2 |
Trisulfur, S3 |
Tetrasulfur, S4 (calc) |
Pentasulfur, S5 (calc) |
|
|
|
|
 |
 |
 |
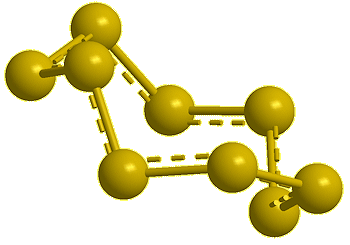 |
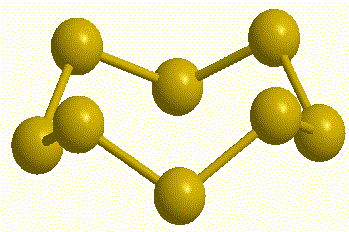
| Cyclo-hexasulfur, S6 |
Cyclo-heptasulfur, S7 (calc) |
Cyclo-octasulfur, S8 |
Cyclo-nonasulfur, S9 (calc) |
|
|
|
|
 |
 |
 |
 |
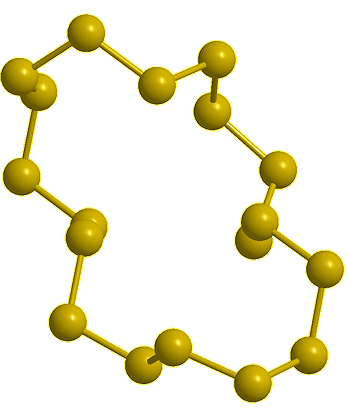
| Cyclo-decasulfur, S10 (calc) |
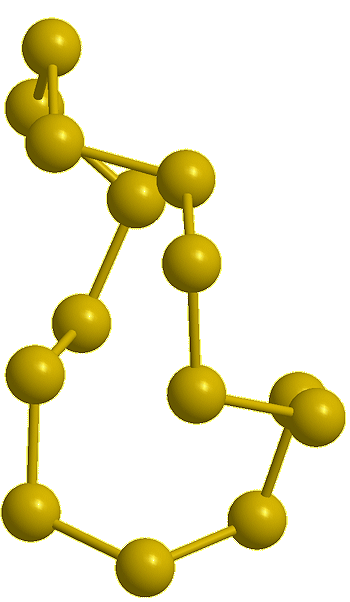
Cyclo-undecasulfur, S11 (calc) |
Cyclo-dodecasulfur, S12 (calc) |
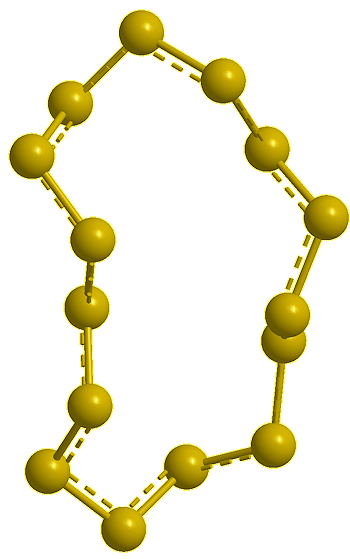
Cyclo-tridecasulfur, S13 (calc) |
|
|
|
|
 |
 |
 |
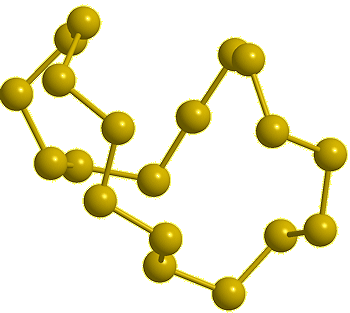 |
| Cyclo-tetradecasulfur, S14 (calc) |
Cyclo-pentadecasulfur, S15 (calc) |
Cyclo-octadecasulfur, S18 (calc) |
Cyclo-icosasulfur, S20 (calc) |
|
|
|
|
Wow, that’s a lot!
Yes, the number of allotropes for sulfur is second only to that of carbon, and this is due to the relatively strong S−S bond (265 kJ mol-1).
Moreover, each allotrope often exists in different polymorphs, which are different crystal structures made up of Sn molecules. Polymorphs are often labelled using Greek letters, α, β, γ, etc.
Do you have an example?
Yes, at room temperature, S8 molecules form brittle, rhombic crystals (known as α-sulfur). When carefully heated to 95.3°C, the rhombic sulfur gradually turns into monoclinic sulfur (β-sulfur). Here, the weak intermolecular forces between the S8 rings are broken and the S8 rings reorganise to form needle-like crystals (β-sulfur).
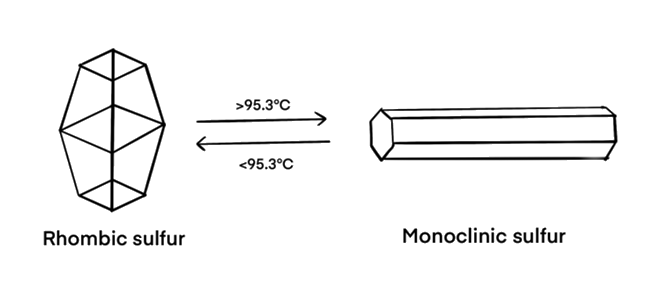
Transition from rhombic sulfur (α-sulfur) to monoclinic sulfur (β-sulfur)
[Image: self-drawn]
Heating β-sulfur above its melting point of 119.6°C breaks the intermolecular forces between the S8 rings, and it liquifies to become dark-orange (λ-sulfur). If the liquid λ-sulfur is heated still further, its viscosity becomes higher, as now the covalent bonds within the S8 rings are broken and the S-fragments link together to form polymers. It reaches its maximum viscosity at around 150°C where the liquid, now almost black in colour, barely flows (μ-sulfur).
However, if the dark-orange treacle-like liquid (λ-sulfur) is poured into cold water to rapidly cool it, the result is ‘plastic sulfur’ (γ-sulfur). Plastic sulfur has a rubber-like property, due to the elasticity caused by the sulfur polymer chains entangling, a bit like spaghetti.
 |
 |
 |
 |
 |
Powdered sulfur
(α-sulfur) |
Dark-orange liquid
λ-sulfur |
Dark molten liquid
μ-sulfur |
Molten plastic
γ-sulfur |
Hardened plastic
γ-sulfur |
What happens when you heat sulfur. Images from: Turning Sulfur Into Plastic?!, Cube Chemistry, Youtube.
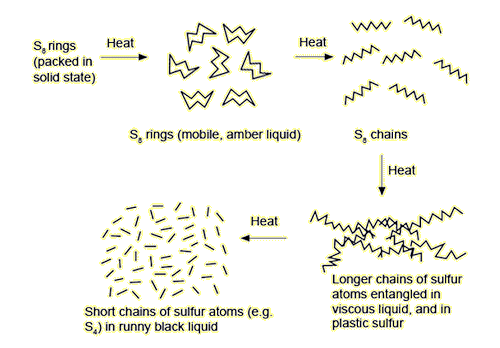
Allotropes of sulfur
[Image: Allotropes of sulfur | Demonstration | RSC Education]
Is that useful?
Yes, very! Sulfur’s ability to form polymer chains is used in the process of ‘vulcanisation’ of rubber, which is essential in all modern tyres.
Vulcanisation? Anything to do with Mr Spock?
No. The term vulcanisation comes from the Roman god of blacksmithing, Vulcan, who forged metal items and weapons using the heat and sulfur in volcanoes.
In 1839, American inventor Charles Goodyear was trying to develop a method to make rubber tyres more durable. At that time, tyres were made from natural rubber, which was brittle when cold but became soft and sticky when hot. After many attempts to harden rubber by simply heating it, he tried mixing rubber with sulfur, but accidentally dropped the mixture into a hot frying pan. Instead of the rubber melting further or vaporising, it remained firm. As the heat was increased, the rubber became harder. By 1844 he had patented this process and had founded the Goodyear tyre company, mass-producing vulcanised rubber tyres on an industrial scale.

Charles Goodyear
[Image: Southworth & Hawes,
Public domain, via Wikimedia Commons] |

A stack of tractor tyres
[Image: By Andy F, CC BY-SA 2.0, via Wikimedia Commons]
|
How did it work?
Heating the sulfur breaks apart the S8 rings to form polymer chains, which then form crosslinks between the long rubber molecules together, a bit like rungs on a ladder. These sulfur crosslinks make the rubber stronger, more durable, and more elastic – perfect for use in tyres.
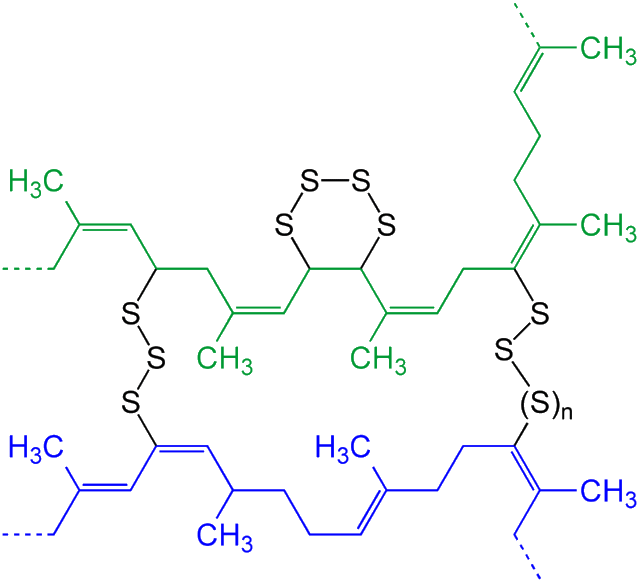
Schematic example of the vulcanization of polyisoprene (a constituent of rubber), with n = 0, 1, 2, 3...
The sulfur chains (black) link together the long polyisoprene molecules (green & blue) making the whole 3D structure stronger.
[Image: Jü, CC0, via Wikimedia Commons]
What happens if you keep on heating sulfur further, i.e. you burn it?
As you saw, heating sulfur causes it to melt into a dark liquid. Heating it further causes it to vaporise, forming diatomic sulfur (S2) molecules in the gas phase. If this vapour is set alight, it burns with a blue flame arising from electronic transitions in excited S2 and SO2 species.
This interesting property occurs on a much larger scale in real life. Sulfur is often found in volcanoes because it's abundant deep within the Earth, and the gases H2S and SO2 readily dissolve in magma. These gases escape as the magma rises and pressure drops. The Indonesian volcano Kawah Ijen is abundant with solid sulfur on its surface and gases of sulfur dioxide and hydrogen sulfide in the magma. When erupted, these gases react together to form elemental sulfur in the form of solid S8 and gaseous S2. The hot S2 emits blue light, making the flames appear blue, which is beautiful at night.
|

Volcano Kawah Ijen erupting at night.
[Image: Thomas Fuhrmann, CC BY-SA 4.0, via Wikimedia Commons] |
What else is sulfur used for?
It’s a vital component in a range of applications from matches, insecticides, and fungicides, but its most common use is to make sulfuric acid – probably the most important chemical used in modern life (see MOTM for May 2008). It is said that almost every item produced in the modern world (clothes, cars, computers, medicines, fertilisers, paints, etc.) have involved the use of sulfuric acid at some point during their fabrication process.
 |
 |
 |
Matches use sulfur
[Image: Yann SEGALEN, CC BY-SA 4.0 via Wikimedia Commons] |
Cartap – a sulfur-based insecticide.
[Image: Ju agrisciences] |
A bottle of sulfuric acid.
[Image: Zoefkris, CC BY-SA 4.0 via Wikimedia Commons]
|
And of course it’s a key component of gunpowder.
Really?
Yes, gunpowder is the earliest known chemical explosive, invented in China around 904 AD. It consists of a mixture of 3 powders: sulfur, charcoal (carbon), and potassium nitrate (saltpeter, KNO3). The sulfur and charcoal are the fuel, while the nitrate is an oxidising agent which decomposes at high temperature to provide oxygen for the reaction. This means that gunpowder still works without atmospheric oxygen. A simplified model of the combustion process is:
2KNO3(s) + S(s) + 3C(s) 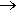 K2S(s) + N2(g) + 3CO2(g) K2S(s) + N2(g) + 3CO2(g)
Sulfur is a key ingredient in gunpowder because it lowers the ignition temperature and increases the burn rate of the mixture.
|

Gunpowder
[Image: Lord Mountbatten, CC BY-SA 3.0 via Wikimedia Commons] |

Fireworks on The Close, Rugby School
[Image: self-taken] |
In fireworks, gunpowder is used as a propellant fuel that burns rapidly to generate gas that lifts the shell into the sky, and burst the shell open at the required altitude.
It’s safe to say that the invention of gunpowder revolutionised the world. Within a relatively short space of time, when the technology reached the west in the 13th century, countries went from fighting with swords and sticks, to guns and cannons.
|
Is sulfur involved with energy storage, like batteries?
Yes. Sulfur (S8) is central to one of the most promising next-generation battery technologies: the lithium–sulfur (Li–S) battery. It is a potential alternative to conventional lithium-ion batteries. Li–S batteries have a very high specific capacity, meaning they can store more energy for a given mass. They are also relatively safer and cheaper to manufacture, as sulfur is abundant and non-toxic.
In a lithium–sulfur battery, S8 is used as the cathode material. During discharge, S8 reacts with lithium to form a series of lithium polysulfides, ultimately producing lithium sulfide (Li2S). When the battery is recharged, these reactions are reversed:
16Li+ + S8 + 16e-  8Li2S 8Li2S
|
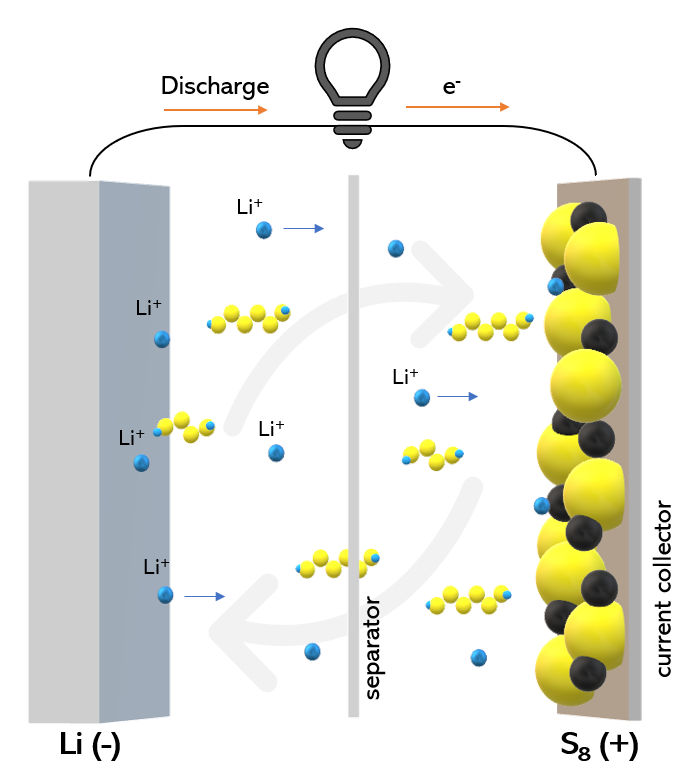
Principle of operation of a lithium-sulfur battery.
[Image: Egibe, CC BY-SA 4.0 via Wikimedia Commons] |
I heard that sulfur is also linked to the origin of life?
Possibly, yes. Underwater volcanoes and hydrothermal vents have been proposed by the 'iron-sulfur world' hypothesis to provide the necessary conditions for biological molecules to form. It’s believed that a few billion years ago, the first metabolic reactions may have not occurred on land, but on iron-sulfide minerals (e.g. FeS, FeS2) on the surface of underwater hydrothermal vents, deep in the prehistoric oceans. These iron sulfide surfaces acted like autocatalysts; at the high temperatures present in the vents, simple inorganic gases (e.g. carbon monoxide, carbon dioxide, hydrogen cyanide and hydrogen sulfide) reacted together to produce the first organic compounds, such as acetates, pyruvates, and simple thioesters. On the other hand, it is also possible that these organic molecules were created inside compartmentalised iron–sulfide structures, which provided protective conditions for these delicate molecules to form. From there, it took another billion years or so to make RNA and DNA - molecules that could replicate themselves - which led to simple cells, and then evolution took over to (eventually) populate the world with living creatures.
|
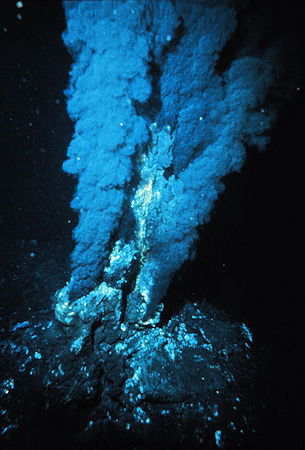
A ‘black smoker’ hydrothermal vent deep in the Atlantic Ocean.
[Image: P. Rona / OAR/National Undersea Research Program (NURP);
NOAA, Public domain, via Wikimedia Commons] |

Io looks yellow because of all the sulfur on its surface.
[Image: NASA / JPL / University of Arizona,
Public domain, via Wikimedia Commons] |
Is sulfur only found on Earth?
Not at all. In fact sulfur is the tenth most abundant element in the universe, and is found in many stars (including our Sun) and planets. It’s been discovered as solid sulfur on Mars and on many moons in the solar system, and as a gas (normally as SO2) in the atmospheres of Venus and some exoplanets. One of the most famous examples is Io, one of the moons of Jupiter, which appears yellow due to the huge deposits of sulfur ejected from its numerous volcanoes. Its atmosphere is about 90% sulfur dioxide.
|

Bibliography
Wikipedia
General sulfur chemistry
- Emsley, J. (2001) Nature's Building Blocks: An A–Z Guide to the Elements. Oxford: Oxford University Press.
- Greenwood, N.N. and Earnshaw, A. (1997) Chemistry of the Elements. 2nd edn. Oxford: Butterworth-Heinemann.
- Royal Society of Chemistry (n.d.) Allotropes of sulfur. Available at: https://edu.rsc.org/experiments/allotropes-of-sulfur/686.article (Accessed: 13 December 2025).
- Rugby School Chemistry (n.d.) Plastic sulfur. Available at: https://www.youtube.com/watch?v=bWY0VZZXJKw (Accessed: 9 December 2025).
- Steudel, R., ed. (2004). Elemental sulfur and sulfur-rich compounds I (Topics in current chemistry). Springer.
Hydrothermal vents
- Martin, W. and Russell, M.J. (2003) 'On the origins of cells: a hypothesis for the evolutionary transitions from abiotic geochemistry to chemoautotrophic prokaryotes, and from prokaryotes to nucleated cells', Phil Trans. Roy. Soc. London. Series B: Biol. Sci., 358(1429), 59–85.
- Wächtershäuser, G. (1988) 'Before enzymes and templates: theory of surface metabolism', Microbiol. Rev., 52(4), 452–484. Available at: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC373159/ (Accessed: 10 December 2025).
Safety of S and its compounds
Sulfur in Climate Change
- Zinke, J. (2019) 'Cool gas in warm summers', Nature Climate Change, 9, 581–584. Available at: https://www.nature.com/articles/s41558-019-0497-6 (Accessed: 10 December 2025).
- Charlson, R.J., Lovelock, J.E., Andreae, M.O. and Warren, S.G. (1987) 'Oceanic phytoplankton, atmospheric sulphur, cloud albedo and climate', Nature, 326(6114), 655–661.
LiS batteries
- Wang, T., Dong, Q., Li, C. & Wei, Z. (2024) ‘Sulfur cathode electrocatalysis in lithium-sulfur batteries: a comprehensive understanding’, Acta Physico-Chimica Sinica, 40(2), 2303061.


 Back to Molecule of the Month page. [DOI:10.6084/m9.figshare.31164610]
Back to Molecule of the Month page. [DOI:10.6084/m9.figshare.31164610]
![]()
![]()
![]()
![]()



































 K2S(s) + N2(g) + 3CO2(g)
K2S(s) + N2(g) + 3CO2(g)



