
![]()
Asphaltene
Road surfaces and more!
![]()
Simon Cotton
University of Birmingham
![]()
Molecule of the Month May 2026
Also available: HTML version.
![]()
 |
AsphalteneRoad surfaces and more!
Simon Cotton
Molecule of the Month May 2026
|
 |
Well, look at the title of this article for clues.
That’s pretty good.
Well, asphaltene isn't just a single molecule, but a group of molecules called asphaltenes. They are black substances found in petroleum (crude oil). They are insoluble in simple alkane solvents, like n-heptane, but dissolve in aromatic hydrocarbons, such as toluene (methylbenzene, see MOTM for July 2022). They are big organic molecules, intermediate in size between polymers and the small molecules usually encountered, with molecular masses usually in the range between approximately 400 and 1500. They contain numerous ring systems, both aromatic rings and heterocyclic rings; these rings often have branches. As well as the non-metals (N, O, S) in the heterocyclic rings, you also find a few metals, notably vanadium and nickel, plus iron.
Asphaltene comes from asphalt (bitumen), which is found naturally where underground oil has risen to the surface forming lakes of asphalt on the surface. One example is the La Brea Tar Pits in Los Angeles (famous for its fossils of mammoths and sabre-toothed tigers) and Pitch Lake in Trinidad.
 |
 |
| The La Brea tar pits in LA. The picture shows a tar pit full of asphalt with a life-size model of a mammoth trapped in the tar. [Image: JERRYE AND ROY KLOTZ MD, CC BY-SA 3.0 via Wikimedia Commons] |
Pitch Lake, Trinidad [Image: Grueslayer, CC BY-SA 4.0 via Wikimedia Commons] |
One good source is the Athabasca oil sands in Alberta (Canada), which contain 15% or more asphaltenes; they are sand and clay containing a black tar. Other sources include the heavy oils of Mexico (Maya oil) and Venezuela. Alberta has more bitumen than the total known world oil reserves, though much is extremely difficult to extract, if not impossible.
 |
 |
| Aerial view of Syncrude Aurora tar sands mine, Athabasca, Canada | Tar sand: oil-impregnated reservoir rocks that have been subjected to long periods of weathering and petroleum degradation, from Alberta, Canada. [Image: James St. John, CC BY 2.0 via Wikimedia Commons] |
Bitumen is the highest-boiling fraction from refining (distillation) of crude oil; asphaltene is the material that gives asphalt in road surfacings their body.
They are generated along with the other components of crude oil, by the decomposition of the molecules in plants and animals, through the effect of temperature and pressure, over geological periods of time.
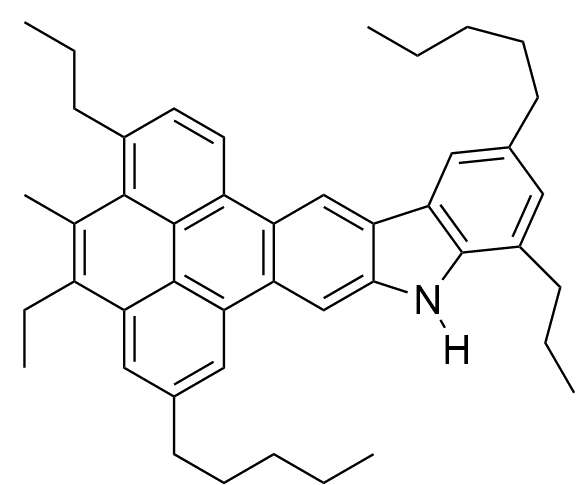
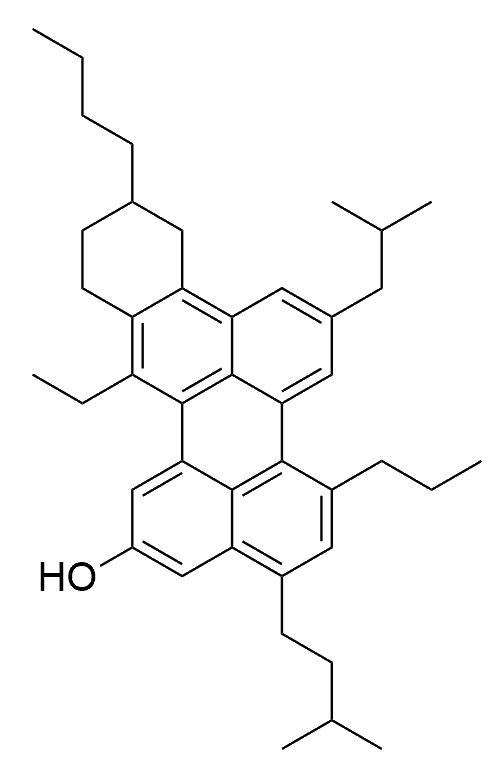
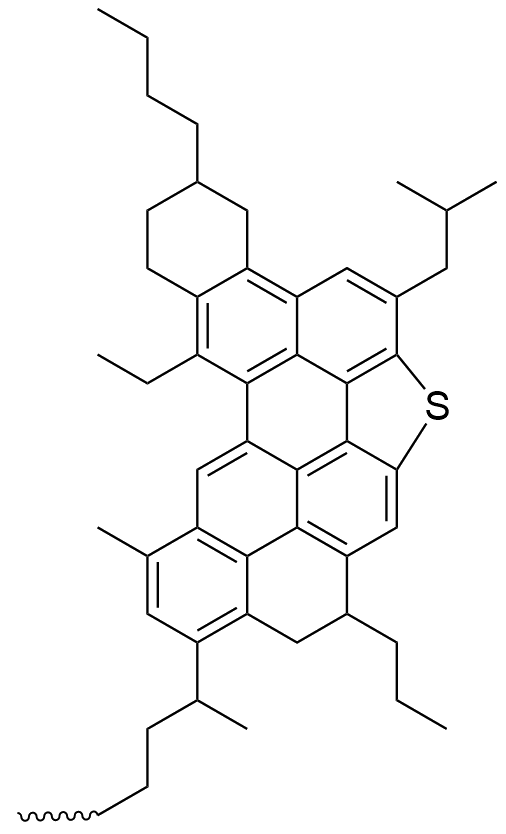
They have complex structures, which are quite difficult to determine, and different asphaltenes have different variations, such as longer or short side-chains, etc. But there are a few general structures, like the example at the top of the page and the two others below, which have more evidence in their favour than others. When there is a central core with sidechains, the structures are called 'island' structures.
 |
 |
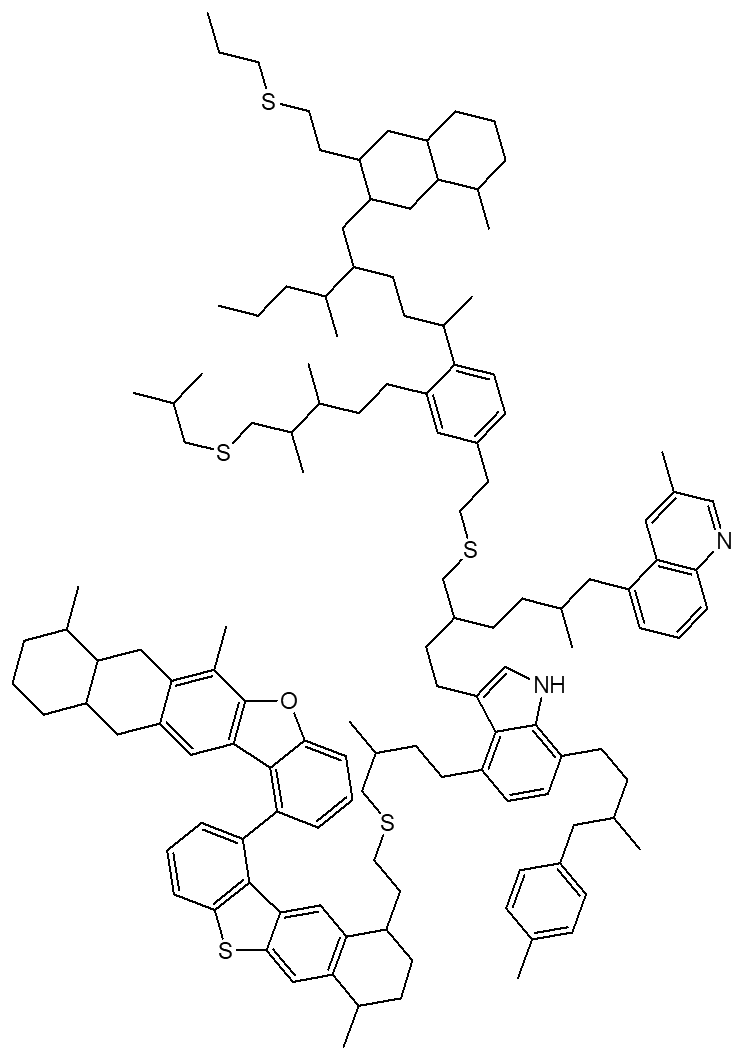
Are all the structures 'islands'?No, a second type has been suggested, like the one shown right, which has fewer rings clustered together, and a more elongated shape. This type is known as an ‘archipelago’ structure. So asphaltenes are a valuable part of crude oil?They are not without their problems. They tend to aggregate, due to strong intermolecular forces between these large molecules. So they contribute massively to the viscosity of crude oil, they can block pipes in refineries and oil wells, as well as oil pipelines. With their low volatility, not only are they hard to ignite, but they also do not burn well. And is there a solution to these problems?Cracking, to produce smaller, fuel-type molecules, is one option (though coke can form in the pores of the catalyst). Recently people have been looking at a variant on cracking. Flash joule heating, which heats asphaltene (or another carbon source) up to above 3,000 K in less than 100 ms, produces high quality graphene. So why bother with it?Well, asphaltenes (usually in the form of bitumen) are used in many building materials, such as road surfacings (tarmac or asphalt, of course!), but also in roofing shingles, and as waterproof coatings. Asphaltenes can be used to treat pollutants and improve water quality by acting as adsorbents for contaminants in water and soil. Their very large surface area allows them to adsorb pollutants onto their surface, and then they can be easily filtered out, leaving the water clean. Also, with available stocks of fossil fuels running lower, people are looking at ‘new’ sources of them, so extraction of asphaltene from the abundant and underexploited oil sands, and converting these to smaller useful hydrocarbons, such as lubricating oils and petrol, may be a real option. |
 |
 |
 |
| An asphalt road. | Laying and flattening asphalt roads |
No need to say thanks, just ‘tar’. [or ‘tar very much’]
![]()
![]()
![]() Back to Molecule of the Month page. [DOI:10.6084/m9.figshare.30166777]
Back to Molecule of the Month page. [DOI:10.6084/m9.figshare.30166777]