![]()
![]()
Molecule of the Month - November 2005
![]()
Also available: Chime Enhanced, VRML and JMol versions.
![]()
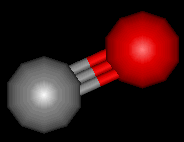
Carbon monoxide is a toxic, colourless and odourless gas. It has been invaluable in helping chemists to extract metals from their ores. However, it must be said that its physical properties make it potentially very dangerous.
Carbon and oxygen can combine to form two gases. When combustion of carbon is complete, i.e. in the presence of plenty of air, the product is mainly carbon dioxide (CO2). Sources of carbon include; coal, coke, charcoal. When combustion of carbon is incomplete, i.e. there is a limited supply of air, only half as much oxygen adds to the carbon, and instead you form carbon monoxide (CO).
 Carbon monoxide is also formed as a pollutant when hydrocarbon fuels (natural gas, petrol, diesel) are burned. The relative amount of CO produced depends on the efficiency of combustion. Older vehicles are checked annually for CO emissions during their MOT test. Interestingly, only one of the two oxides of carbon, doesn't support combustion and it is for this reason that carbon dioxide is used in fire extinguishers. Carbon monoxide does support combustion and burns with a pale blue flame. The blue flame used to be seen over the fires made from coke (essentially a very pure form of carbon) by night watchmen on industrial sites.
Carbon monoxide is also formed as a pollutant when hydrocarbon fuels (natural gas, petrol, diesel) are burned. The relative amount of CO produced depends on the efficiency of combustion. Older vehicles are checked annually for CO emissions during their MOT test. Interestingly, only one of the two oxides of carbon, doesn't support combustion and it is for this reason that carbon dioxide is used in fire extinguishers. Carbon monoxide does support combustion and burns with a pale blue flame. The blue flame used to be seen over the fires made from coke (essentially a very pure form of carbon) by night watchmen on industrial sites.
2 CO (g) + O2 (g) ![]() 2 CO2 (g)
2 CO2 (g)
When making carbon monoxide a source of carbon dioxide gas is needed. This could be from a CO2 cylinder or even dry ice (solid CO2). If neither is available carbon dioxide could be generated by the neutralisation reactions between an acid and a carbonate or an acid and a hydrogen carbonate.
2HCl (aq) + CaCO3 (s) ![]() CaCl2 (aq) + H2O (l) + CO2 (g)
CaCl2 (aq) + H2O (l) + CO2 (g)
HCl (aq) + NaHCO3 (s) ![]() NaCl (aq) + H2O (l) + CO2 (g)
NaCl (aq) + H2O (l) + CO2 (g)
When carbon dioxide gas is passed over heated charcoal it forms carbon monoxide.
CO2 (g) + C (s) ![]() 2CO (g)
2CO (g)
There will also be unreacted carbon dioxide, which needs to be removed. Carbon dioxide is removed by reacting it with an aqueous solution of sodium hydroxide.
2 NaOH (aq) + CO2 (g) ![]() Na2CO3 (aq) + H2O (l)
Na2CO3 (aq) + H2O (l)
Another convenient way to prepare carbon monoxide is the dehydration of methanoic acid using conc. H2SO4.
HCOOH (aq) ![]() CO (g) + H2O (l)
CO (g) + H2O (l)
Dehydration of a methanoic salt such as sodium methanoate also works well. In this case you dribble concentrated sulphuric acid directly onto the solid. The carbon monoxide, which evolves, can be collected under water. Methanoic acid is found in nettles and ants.
 Carbon monoxide is a very poisonous gas. It is poisonous at levels of only 0.1% (1000 ppm). Its toxicity arises from its ability to bind to transition metals such as iron found at the centre of a haem molecule. Carbon monoxide is attracted to haemoglobin over 200 times more strongly than oxygen. Therefore, in the blood, the presence of carbon monoxide prevents some of the haemoglobin found in red blood cells from carrying sufficient oxygen.
Carbon monoxide is a very poisonous gas. It is poisonous at levels of only 0.1% (1000 ppm). Its toxicity arises from its ability to bind to transition metals such as iron found at the centre of a haem molecule. Carbon monoxide is attracted to haemoglobin over 200 times more strongly than oxygen. Therefore, in the blood, the presence of carbon monoxide prevents some of the haemoglobin found in red blood cells from carrying sufficient oxygen.
This fact is certainly worth considering if you should be tempted to smoke a cigarette. Smokers have been found to have quite high levels of carbon monoxide in their blood, long after they have finished lighting up their chosen cigarette.
 Symptoms of carbon monoxide poisoning are dizziness and headaches. These sufferings can be confused with other illnesses such as influenza. Carbon monoxide poisoning can be recognised, as victims will often have unnaturally bright red lips.
Symptoms of carbon monoxide poisoning are dizziness and headaches. These sufferings can be confused with other illnesses such as influenza. Carbon monoxide poisoning can be recognised, as victims will often have unnaturally bright red lips.
Prolonged exposure to carbon monoxide can eventually lead to death. Carbon monoxide has been used as the poison in suicides. Even more disturbing was the use of carbon monoxide by Nazis in WWII to kill its victims of the death camps. More recently, there have been cases of rogue landlords not properly maintaining gas appliances, which led to the death of tenants often students. The law in the UK now requires the annual checking of boilers, gas cookers, and gas fires by registered engineers. The following equation shows what happens when there is incomplete combustion of natural gas, which is primarily methane.
2 CH4 (g) + 3 O2 (g) ![]() 2 CO (g) + 4 H2O (g)
2 CO (g) + 4 H2O (g)
 Before vast quantities of natural gas were discovered beneath the seas and oceans, we used to burn coal gas. Coal gas was produced when coal was heated in the absence of air. Its main components are hydrogen methane, and carbon monoxide.
Before vast quantities of natural gas were discovered beneath the seas and oceans, we used to burn coal gas. Coal gas was produced when coal was heated in the absence of air. Its main components are hydrogen methane, and carbon monoxide.
Carbon monoxide sometimes occurs in coal mines. At one time canaries were taken down in mines to detect poisonous gases. Canaries would be killed at doses not quite lethal to miners. Today gases are detected more humanely with instruments.
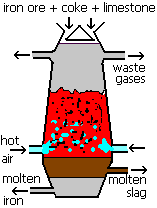 The first time most students meet carbon monoxide in a chemistry lesson is in its use within the Blast Furnace. The Blast furnace is how iron is extracted from its ore, haematite (iron(III)oxide Fe2O3).
The first time most students meet carbon monoxide in a chemistry lesson is in its use within the Blast Furnace. The Blast furnace is how iron is extracted from its ore, haematite (iron(III)oxide Fe2O3).
Fe2O3 (s) + 3 CO (g) ![]() 2 Fe (l) + 3 CO2 (g)
2 Fe (l) + 3 CO2 (g)
Carbon monoxide is a strong reducing agent and reduces metal oxides for metals less reactive than carbon. The following table is useful for the different definitions of reduction. As oxidation is the opposite of reduction you only need to learn half the facts!
| Reduction | Oxidation |
|---|---|
| Loss of oxygen | Gain of oxygen |
| Gain of hydrogen | Loss of hydrogen |
| Gain of electrons | Loss of electrons |
| Decrease in O.N. | Increase in O.N. |
Several gases (H2, CH4 and CO) have historically been used as reducing agents. One definition of a reducing agent I particularly like is to think of it as being an oxygen grabber. It is important to remember that the reducing agent itself gets oxidised. A quick and simple laboratory reduction can be achieved by heating a mixture of black copper(II)oxide with carbon powder in a test tube. After several minutes of heating the reddish coloured copper can be seen on the side of the test tube. Essentially carbon acts as a reducing agent as well as the carbon monoxide that is inevitably formed by its heating in air. The following reactions are all occurring in this simple experiment.
CuO (s) + CO (g) ![]() Cu (s) + CO2 (g)
Cu (s) + CO2 (g)
CuO (s) + C (s) ![]() Cu (s) + CO (g)
Cu (s) + CO (g)
C (s) + O2 (g) ![]() 2 CO (g)
2 CO (g)
C (s) + O2 (g) ![]() CO2 (g)
CO2 (g)
CO2 (g) + C (s) ![]() 2 CO (g)
2 CO (g)
Carbon monoxide does not show acidic or basic properties. Its feeble Lewis acidity is seen through its formation of H3BCO with borane (BH3). Carbon monoxide has a remarkable affinity for transition metals (located between Groups 2 and 3 of the Periodic Table). The first examples of metal carbonyls was back in 1888, when tetracarbonyl nickel (0) Ni(CO)4 and pentacarbonyl iron (0) Fe(CO)5 were prepared and characterised. The former complex forms part of the Mond Process for the purification of Nickel. Ni(CO)4 is distilled to give pure nickel.
Ni (s) + 4 CO (g) ![]() Ni(CO)4
Ni(CO)4 ![]() Ni (s) + 4 CO (g)
Ni (s) + 4 CO (g)
Carbon monoxide is so reactive with nickel that within a couple of minutes it will have etched the surface. Ni(CO)4 is highly toxic with a musty smell. As well as being flammable this tetrahedral complex decomposes easily into its constituents. Carbon monoxide is acting as a ligand towards the transition metal through the lone pair on the carbon atom. Diatomic carbon monoxide has a triple bond between its atoms. One of the bonds forming the triple bond is a dative covalent bond.
 Please try this at home
Please try this at homeDetecting carbon monoxide is a simple task these days. Most hardware stores sell special spots impregnated with palladium compounds, which darken on exposure to carbon monoxide.
![]()
![]() Back to Molecule of the Month page. [DOI:10.6084/m9.figshare.5436793]
Back to Molecule of the Month page. [DOI:10.6084/m9.figshare.5436793]