![]()
DOXYCYCLINE
(and other tetracyclines)
The antibiotic that's an alternative to penicillin
![]()
Simon Cotton
Uppingham School, Rutland, UK
![]()
Molecule of the Month September 2011
Also available: JSMol version.
![]()
DOXYCYCLINE(and other tetracyclines)The antibiotic that's an alternative to penicillin
Simon Cotton
Molecule of the Month September 2011
|
 |
 Why is it important?
Why is it important?In the wake of the attacks on the Twin Towers in New York on 11th September 2001, several letters containing anthrax spores were sent to two U. S. Senators and to various media outlets. Five people died of inhalational anthrax and seventeen others were taken ill. Bruce Edwards Ivins, a scientist at the U. S. government's bio-defence laboratories at Fort Detrick, Maryland, came under suspicion, and he committed suicide on July 27th 2008.
The anthrax outbreaks led to people suspected of exposure to anthrax being given the drug Cipro (Ciprofloxacin) but Doxycycline was recommended as an equally effective and cheaper alternative. Doxycycline (vibramycin) is effective against several diseases, including Yersinia pestis (bubonic plague), Lyme disease and Rocky Mountain spotted fever, as well as anthrax.
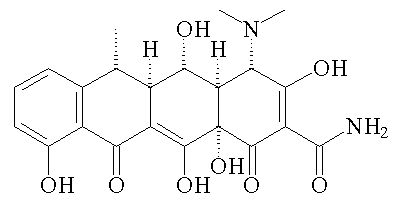
Doxycycline is synthetic, but some tetracyclines are produced naturally by Streptomyces bacteria, such as chlorotetracycline (Aureomycin), which was the first one to be discovered; it was isolated from Streptomyces aurofaciens obtained from samples of Missouri soil in 1945 and marketed as a medicine in 1948.
Chlorotetracycline (Aureomycin)
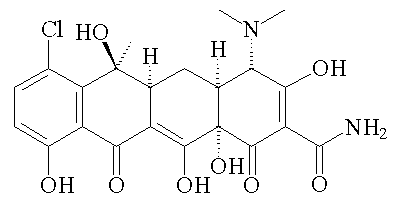
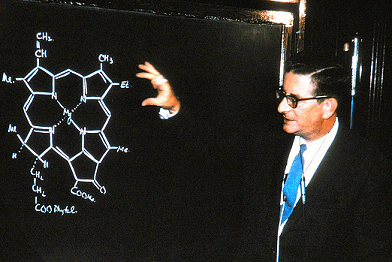 In 1952 it was found that you could hydrogenate chlorotetracycline under mild conditions (H2; Pd/C catalyst) and produce tetracycline itself. Pfizer isolated oxytetracycline from Streptomyces rimosus in the soil near their Indiana plant. A research team led by the great R.B. Woodward (photo, right) of Harvard went to work to solve the structure and published it in 1953.
In 1952 it was found that you could hydrogenate chlorotetracycline under mild conditions (H2; Pd/C catalyst) and produce tetracycline itself. Pfizer isolated oxytetracycline from Streptomyces rimosus in the soil near their Indiana plant. A research team led by the great R.B. Woodward (photo, right) of Harvard went to work to solve the structure and published it in 1953.
Another Nobel laureate, D.H.R. Barton, said that "The most brilliant analysis ever done on a structural puzzle was surely the solution (1953) of the terramycin problem."
At that time, a 'gold rush' was on to discover antibiotics from natural sources.
Chlorotetracycline had advantages over the other antibiotics that were available at the time (including penicillin) as it could be taken orally and was also effective against both Gram-positive and Gram-negative bacteria. By that time, resistance to penicillin had already been noted, so alternative antibiotics were a must.
In 2005, Andrew Myers' group at Harvard University described an 18-step synthesis, starting from benzoic acid.
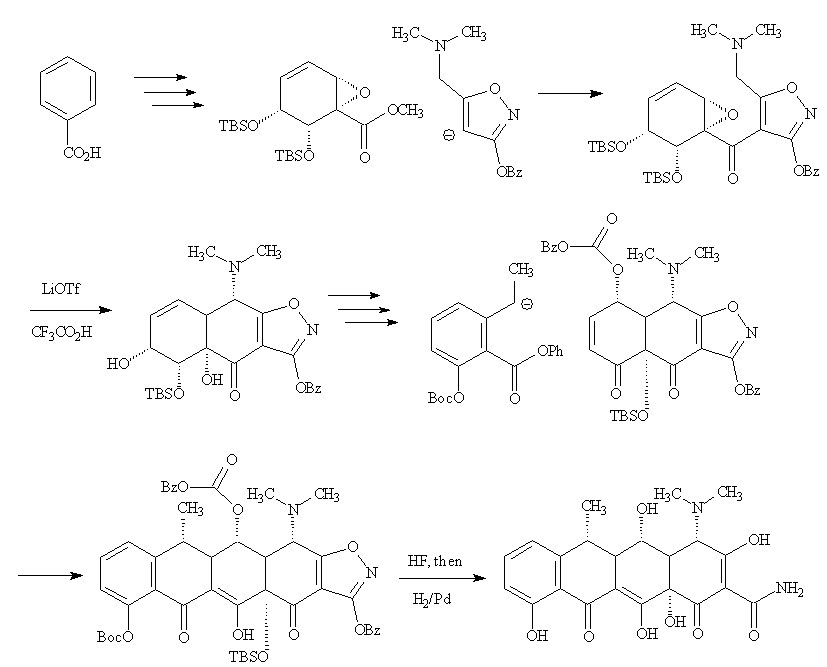
The benzoic acid molecule forms the basis of the B ring of the doxycycline, around which the A, C and D rings are added by various ring-forming reactions. First the benzoic acid undergoes microbial dihydroxylation, followed by further steps including epoxidation and the introduction of protecting tert-butyldimethylsilyl groups (TBS) to afford an enantiomerically-pure epoxyester. A deprotonated dimethylamino-substituted isoxazole is then added, subsequent ring closure in the successive reactions with lithium triflate generating the A ring, and trifluoroacetic acid removing a TBS group. The C and D rings were then added by means of a coupling reaction with another carbanionic reagent via a Michael-Dieckmann sequence. Removal of the protecting groups (TBS = tert-butyldimethylsilyl; BOC = tert-butyloxycarbonyl, COtC4H9) afforded doxycycline, stereoselectively. The yield in this virtuoso demonstration of synthetic organic chemistry was 8.3%.
Tetracyclines inhibit bacterial protein synthesis. They bind to the 16S part of the 30S subunit in the ribosome, and interfere with the binding of amino-acylated tRNA, stopping the messenger-RNA codon reading the t-RNA anticodon. This is a key step of protein synthesis, and means that new aminoacids cannot be added to the nascent polypeptide chain. This explains why tetracyclines are effective against a wide range of both gram-positive and gram-negative bacteria.
 The tetracyclines are not used as widely as they once were, because of the emergence of resistant strains of bacteria. In part, this is due to the use of small amounts of antibiotics in animal feeds of animals such as pigs, chicken and cattle, in order to control disease and promote growth.
The tetracyclines are not used as widely as they once were, because of the emergence of resistant strains of bacteria. In part, this is due to the use of small amounts of antibiotics in animal feeds of animals such as pigs, chicken and cattle, in order to control disease and promote growth.
Apart from doxycycline, several tetracyclines still find wide application. Many members of the tetracycline family have been made by semi-synthesis, using a naturally produced molecule to provide the core, then modifying its structure. Among the newer tetracyclines, tigecycline (Tygacil; 2005) is active against resistant bacteria such as Staphylococcus aureus.
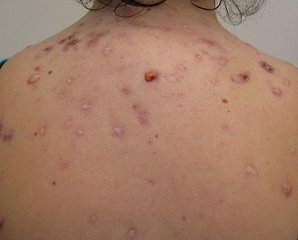
Like other tetracyclines, minocycline is used as an oral antibiotic for acne (picture, right), but in 2011 Canadian scientists carried out a screening of combinations of minocycline with other drugs and showed that a combination of minocycline with the well-known anti-diarrhoea drug loperamide was a potent antibacterial. Tests have not been carried out on humans yet, but it is possible that such combinations could give new life to tetracyclines in medicine.
Recently Andrew Myers' group have synthesised five-ring pentacyclines that show promise against pathogens that are resistant to some other medications.
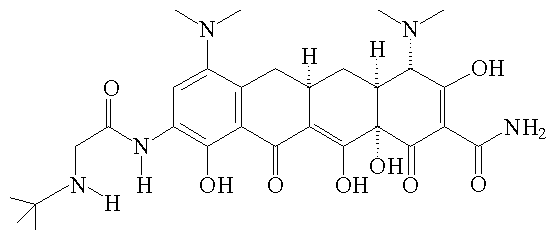 |
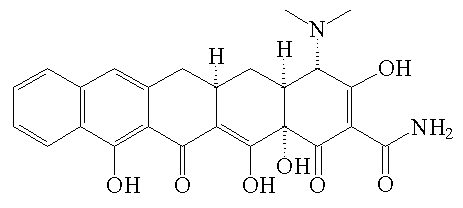 |
Tigecycline |
Pentacycline |
Recently doxycycline has been widely used as malaria prophylaxis. It might cause a bit of nausea when you start taking it (starting at half the dose is the way round this) but it's a lot safer than some of the alternatives and, as an added bonus, you get a better suntan as a result too!
 But tetracyclines have actually been used for over 1500 years. It turns out that tetracycline was present in ancient Nubian beer and contributed to low rates of infectious disease.
But tetracyclines have actually been used for over 1500 years. It turns out that tetracycline was present in ancient Nubian beer and contributed to low rates of infectious disease.
The Nubian beer wasn't like the present-day stuff; it was more like a syrupy gruel. 30 years ago, scientists at Emory University detected a green fluorescence when UV light was shone on bones from Nubian skeletons, dating from between 350 and 550 AD (see photo, below). This indicated the presence of tetracycline.
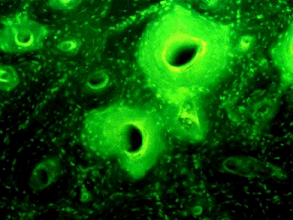
Tetracycline Nubian bone fluorescence
(Reproduced by permission of Professor George J. Armelagos.)
It was found that the beer was made by fermenting grain which contained streptomyces bacteria, found in soil. Further research indicated there were high levels of tetracycline in the bones of even small children. The ancient Nubians recognised the health effects of the beer and deliberately produced it, even if they did not know about tetracycline.
Acknowledgements Thanks are due to Professor George Armelagos of Emory University to reproduce an illustration, and to Professor James Anderson of UCL for advice.
![]()
Chapman and Hall Combined Chemical Dictionary compounds code numbers: tetracycline BDQ00-F; chlorotetracycline CKR88-Y; oxytetracycline CLB98-G; doxycycline BDT97-C; tigecycline KKB19-M; minocycline CKJ08-M (props, bibliography)
![]()
![]() Back to Molecule of the Month page. [DOI:10.6084/m9.figshare.5255548]
Back to Molecule of the Month page. [DOI:10.6084/m9.figshare.5255548]