![]()
Ethene
a.k.a. Ethylene
![]()
Simon Cotton
Uppingham School, Rutland, UK
![]()
Molecule of the Month December 2006
Also available: JSMol version.
![]()

Ethenea.k.a. Ethylene
Simon Cotton
Molecule of the Month December 2006
|
 |
It happened 2500 years ago.
Delphi, on the slopes of Mount Parnassus in Greece.
The same, the woman who sat in a small chamber under the shrine to communicate with Apollo.
Most famously in 480 BC, Xerxes, the Persian emperor, was returning to Greece with a huge army and fleet to avenge his father Darius I, whom the Greeks had defeated at the Battle of Marathon (490 BC). The Athenians sent a delegation to ask the Oracle for guidance, only to be insulted and turned away with predictions of doom. They returned more humbly, to be told that Athens would be destroyed, but that Zeus would grant the goddess Athena, protector of Athens, a "wooden wall" that would be uncaptured, and that Salamis would be the death of many a woman's son. Debate raged among the Greeks whether this meant a wooden wall round the city or the Greek fleet. At the urging of Themistocles, they reinforced their fleet. And so the Athenian fleet defeated the much larger Persian fleet at Salamis, as Xerxes, powerless to intervene, watched from his golden throne, high on the slopes of Mount Aegaleus. And Aeschylus wrote "The Persians", the earliest surviving European historical play.
 So where do the hydrocarbons come in?
So where do the hydrocarbons come in?Ethene has been detected in local spring waters; Plutarch (ca. 46-120 AD) referred to her inhaling sweet smelling vapours, and ethene is a sweet smelling hydrocarbon. It also has anaesthetic properties.
Writing around 430 BC, the ancient historian Herodotus related that an earthquake at Delphi had deterred the Persian army from robbing the shrine in 480 BC. Modern geologists have now located fault lines intersecting below the shrine. The local rocks contain bitumen; friction between moving rocks would generate the heat necessary for a cracking reaction that would break bitumen into smaller hydrocarbon molecules such as ethene. Other small hydrocarbon molecules such as methane and ethane have also been found in these rocks and waters.
Ethene has been employed as an anaesthetic, though it is not used nowadays, since it is flammable and also causes cardiac arrhythmogenicity.
One theory is that it dissolves in the nerve cell membranes, affecting the transmission of nerve messages. Others think that they can interact with proteins around the mouths of channels in the membranes.
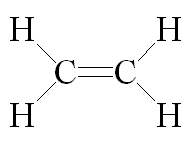
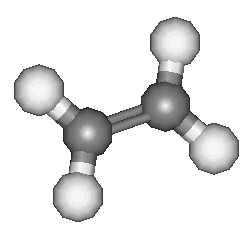
It is one of those rare flat molecules, like benzene; the H-C-H bond angle is 117°. It boils at about -104°C, so at room temperature ethene is a colourless gas with a faintly sweet smell and taste.
 |
 |
 |
Ethylene was the original name, applied in the mid-19th century, showing it was derived from "ethyl", C2H5-. It was only late in the 19th century that the simpler systematic name of ethene started to be adopted, but by then the chemical industry was used to calling it ethylene. It still does. Industry makes ethene in very large quantities by cracking alkanes.
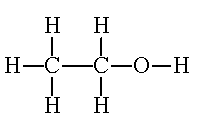
Certainly not. The double bond in ethene, which makes that part of the molecule electron-rich, causes it to be more reactive than an alkane. This is why it does not survive in nature (as alkanes do) but it is very valuable to the petrochemical industry for making many other chemicals, such as polythene, ethanol (solvent and fuel), ethane-1,2-diol for antifreeze and making polymers, styrene, from which polystyrene is made, and vinyl chloride, source of PVC. And it is an important raw material in the Wacker process.
 |
 |
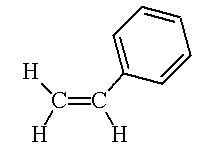 |
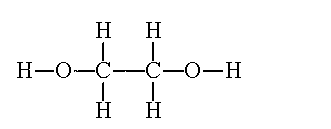
| Ethanol | Ethane-1,2-diol | Styrene |
The introduction of polythene revolutionised packaging and wrapping, superseding paper bags, as it had a big advantage in its wet strength (the downside being its lack of biodegradability).
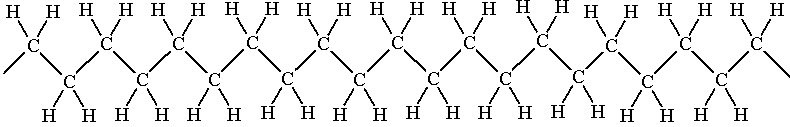 |
| Polythene |
Polythene's discovery on Monday 27th March 1933 was an accident. Two chemists at ICI, Reginald Gibson and Eric Fawcett, had reacted ethene with benzaldehyde, hoping to insert ethene into the C-H bond of the aldehyde group and make ethyl phenyl ketone, CH3CH2COC6H5. They were operating at 2000 times atmospheric pressure and 180°C in an open laboratory (risk assessment ? the mind boggles) and when the experiment did not explode, they got a white waxy solid. They had trouble reproducing the product, and eventually it was realised that adventitious traces of oxygen acted as a catalyst for the polymerisation.
Polythene can be made by two main routes, which give slightly different products. High-temperature, high-pressure polymerisation (the original method) gives low-density polythene (LDPE), which has quite a bit of chain branching, so the chains do not pack well. This makes it rather soft and weak, so it tends to be used for making wrappings and bags, applications that do not require strength. Synthesis by the Ziegler-Natta method, which employs organometallic catalysts at room temperature, gives high-density polythene (HDPE); there is less chain branching, so it is stronger, and tends to find applications like washing up bowls and milk bottles. The two forms have different recycling symbols.
There are several other forms. Anything to do with Polythene Pam?Yes, and this shows how widely used polythene was by the late 60s. The Beatles' Abbey Road track (1969) may have been inspired by a fan who ate polythene. |
Polythene Pam (Lennon/McCartney) "Well you should see Polythene Pam Get a dose of her in jackboots and kilt |
The Wacker (or Hoechst-Wacker) process was developed by German chemists as a commercial method of oxidising alkenes to carbonyl compounds. Ethene gets converted to ethanal (acetaldehyde) in a reaction using a PdCl2 catalyst, but copper(II) chloride, hydrochloric acid and oxygen all play an important role in returning the catalyst to its original state and in thus maintaining the catalytic cycle.
[PdCl4]2- + C2H4 + H2O ![]() CH3CHO + Pd + 2H+ + 4Cl-
CH3CHO + Pd + 2H+ + 4Cl-
Pd + 2CuCl2 + 2Cl- ![]() [PdCl4]2- + 2CuCl
[PdCl4]2- + 2CuCl
2 CuCl + �O2 + 2H+ + 2Cl- ![]() 2CuCl2 + H2O
2CuCl2 + H2O
 Anything else about ethene?
Anything else about ethene?In nature, ethene can be produced in cells from the amino acid methionine. It has an important role as a plant growth hormone. It is produced by ripening fruit and it also brings fruit to ripeness. Nowadays fruit like bananas are picked unripe, stored until they are needed for the market, then exposed to ethene to bring them to ripeness. Similarly, if green tomatoes are placed in a plastic bag with a ripening banana, then they go red. This is why a ripe fruit (which produces ethene) should not be stacked with unripe fruit, as "one rotten apple spoils the barrel".
The explosive nature of mixtures of ethene with air was one reason for the use of ethene as an anaesthetic being discontinued. Sadly, a French professor's death in an explosion at the University of Mulhouse on Friday 24th March 2006 has been blamed on an ethene explosion.
![]()
![]()
![]() Back to Molecule of the Month page. [DOI:10.6084/m9.figshare.5248996]
Back to Molecule of the Month page. [DOI:10.6084/m9.figshare.5248996]