![]()
 |
KevlarThe super-tough fibre used |
![]()
Paul May
University of Bristol
![]()
Molecule of the Month November 2010
Also available: HTML version.
![]()

 |
Paul May
Molecule of the Month November 2010
|
 |
Kevlar is well known as the material from which bullet-proof vests and body-armour is made. But it can be used in many more applications, such as bicycle tyres, yacht sails, ropes, and brakepads. It can do this because, despite it being a fibre, it has an extremely high stength-to-weight ratio, often qiuoted as being 5 times stronger than an equal weight of steel. It is actually a trademark of the chemical company DuPont, who first synthesised it in 1965.
 Who invented it?
Who invented it?A chemist called Stephanie Kwolek invented Kevlar while working for DuPont in their Wilmington labs. In anticipation of a gas shortage, in 1964 the DuPont research group led by Kwolek began searching for a new lightweight but strong fibre to use for tyres. One day in 1965, while trying to dissolve one of her polymers, something strange happened. She reported:
"Ordinarily a polymer solution sort of reminds you of molasses, although it may not be as thick. And it’s generally transparent. This polymer solution poured almost like water, and it was cloudy. I thought, ‘There’s something different about this. This may be very useful.'"
Normally such a "cloudy, opalescent, and low viscosity" solution would just be thrown away. However the technician, Charles Smullen, who ran the "spinneret" persuaded Kwolek to test her solution. When spun into a fibre, they were amazed to find that, unlike the other well-known fibre of the time, nylon, this new fibre did not break. Both her supervisor and laboratory director understood the significance of her discovery and a new field of aramid polymer chemistry was born. It then took six years to produce Kevlar commercially, finally reaching the market in 1971.
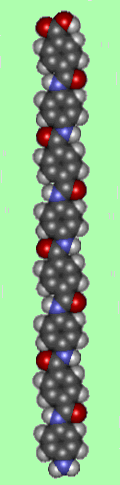
What actually is it?Kevlar is a type of fibre called an aramid, short for 'aromatic polyamide', in which the molecules form long, highly-oriented chains. The fibres can be spun or woven into mats or fabrics to exploit these exceptional properties. Kevlar is made by a condensation reaction of an amine (1,4-phenylene-diamine) and and acid chloride (terephthaloyl chloride).
A section of an individual kevlar chain looks like:
The kevlar chains are relatively rigid and tend to form mostly planar sheets, similar to those of silk. This is due to the para-orientation of the benzene rings. When kevlar is spun the chains lock together via H-bonds to form a sheet that has a very high tensile strength. The sheets also stack radially, like the spokes on a wheel, allowing additional interactions between the face-to-face aromatic groups on neighbouring sheets to help to increase the strength of the overall fibre.
|
Kevlar has some superb properties:
 Can it be used for other things?
Can it be used for other things?As mentioned above, the most well-known application for kevlar is in personal armour, such as bulletproof vests, combat helmets, face-masks, etc. But Kevlar can also be used in sports equipment, such as inner linings for bicycle tyres, tabletennis bats, bow strings for archery, paraglider suspension lines, and motorcycle safety clothing. It can also be used in snare drums, since it can be pulled very taut leading to a cleaner sound. Kevlar fibres can be woven into ropes or cables, which have been used to support suspension bridges. It is also used as a protective outer sheath for optical fibres. Kevlar has been used as a replacement for the asbestos in automobile brake pads, and in some rubber hose joints. Kevlar is often used in conjunction with glass fibre and carbon fibres as reinforcing agents in composite materials. Such composites find uses in aerospace, racing cars (like the kevlar Corvette in the photo, right), helicopter rotor-blades, and many specialised pieces of sports equipment.
![]()
![]()