![]()
METHANE
(Natural gas)
![]()
Simon Cotton
Uppingham School, Rutland, UK
![]()
Molecule of the Month December 2007
Also available: HTML version.
![]()

METHANE(Natural gas)
Simon Cotton
Molecule of the Month December 2007
|
 |
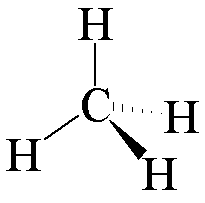
It's meth-ane, not 'me'-thane. It is the smallest hydrocarbon and the lightest stable organic molecule. A methane molecule has a regular tetrahedral structure (Td symmetry), its H-C-H angles are all 109½°.
 |
 |
|
| Methane | ...and its spacefill representation. |
It is not very soluble in water on account of its non-polar nature. It is a colourless, odourless gas at room temperature (m.p. 90 K, b.p. 109 K) and is less dense than air.
Because methane is odourless, natural gas supplied for domestic use has a trace of an obnoxious organic sulphur compound such as Me3CSH or Me-S-Me deliberately added for ease of detecting any leak.
 The combustion of methane is a very exothermic reaction (ΔH = -891 kJ/mol) and on that account methane is widely used as a fuel, not least because the volatile and non-toxic products make it "clean", though one product, carbon dioxide, is a "greenhouse gas".
The combustion of methane is a very exothermic reaction (ΔH = -891 kJ/mol) and on that account methane is widely used as a fuel, not least because the volatile and non-toxic products make it "clean", though one product, carbon dioxide, is a "greenhouse gas".
CH4 (g) + 2O2 (g) ![]() CO2 (g) + 2H2O (g)
CO2 (g) + 2H2O (g)
Incomplete combustion yields carbon (soot) or toxic carbon monoxide.
Methane is the main component of "natural gas", which contains between 70 and 95% methane, often over 90%. This can be purified to remove other low-boiling components like noble gases and slightly larger alkanes, then condensed at around -160°C as liquefied natural gas (LNG). This is a much more compact way of storing methane, and when pipelines are not available, it can be transported in cryogenic tankers by sea or road.
 So, North Sea Gas is largely methane
So, North Sea Gas is largely methaneIndeed. North Sea Oil (and gas) was discovered in the early 1960s, with first supplies being piped ashore in the UK in 1975. The resource is shared between the UK, Norway, Denmark, Germany and the Netherlands, mainly the first two. Production peaked in 1999. This enabled the UK to become - briefly - an exporter of energy and also enabled the Conservative Government of the 1980s and early 1990s to move away from dependence upon coal-fired power stations towards the use of gas as a fuel (the "Dash for Gas"). The UK is now a net importer, looking to obtain supplies from sources including Russia.
Its reaction with steam in the presence of a hot nickel catalyst affords the mixture of CO and H2 known as "synthesis gas", widely used to make chemicals such as hydrogen, methanol and ethanoic acid, for example.
CH4 (g) + H2O (g) ![]() CO (g) + 3H2 (g)
CO (g) + 3H2 (g)
Billions of years ago, volcanic activity was responsible for large amounts of methane in the Earth's atmosphere; subsequently the first primitive bacteria synthesised methane from CO2 and hydrogen, and methane levels did not fall until the arrival of the first organisms to photosynthesise, which generated oxygen. The concentration of methane in the Earth's atmosphere is quoted as 1.75 ppb in 1998.
Nowadays, the main source of methane is "Natural Gas", which originates in the same way as crude oil, by the anaerobic decomposition of microscopic sea animals under the pressure of layers of silt and mud over a period of millions of years. This can also occur in swamps and marshes, when plant matter decays ("marsh gas"), and in landfill sites; in digesters breaking down sewage sludge; and by enteric fermentation in livestock (e.g. ruminants such as cattle and sheep), as well as being formed when termites digest the cellulose in the wood they have eaten.

John Dalton (1766-1844), collecting marsh gas (methane) with youngsters near Manchester.
Methane can be made in the laboratory by a variety of reactions, including the action of water and aluminium carbide (which contains discrete C4- ions) :
Al4C3 (s) + 12H2O (l) ![]() 3CH4 (g) + 4Al(OH)3 (s)
3CH4 (g) + 4Al(OH)3 (s)
as well as by heating a mixture of solid sodium hydroxide with sodium ethanoate:-
CH3COONa (s) + NaOH (s) ![]() CH4 (g) + Na2CO3 (s)
CH4 (g) + Na2CO3 (s)
 And methane is a "greenhouse gas"?
And methane is a "greenhouse gas"?Methane is classed as a greenhouse gas, and its Global Warming Potential (GWP) calculated over a 100-year span is 22-23 times greater than that of CO2. Because of this, there is concern about methane emissions from diverse natural sources, such as mud volcanoes and ruminants, together with the possibility of losses from methane hydrates, released as the oceans warm up. 1990 estimates put "natural" methane emissions at around 155 million metric tons a year; at that time methane emissions from "man made" sources came to 350 million metric tons a year.
It has recently (2006) been suggested that trees and other green plants in aerobic environments are responsible for adding large amounts of methane to the Earth's atmosphere, but another group, using 13C-labelling to distinguish emissions from plants from 12CH4 "background" in the atmosphere, has found no evidence for this.
When they are under pressure, as under cold sediments at the bottom of oceans, methane molecules get locked up inside cavities within the structure of ice, forming what is known as known as methane clathrate (other alkane molecules are larger, and cannot fit into the cavities). This methane is largely formed by anaerobic bacterial breakdown of organic matter. Since methane clathrates have been estimated to store more energy than the total fossil fuel reserves of the whole world, there has been interest in harvesting this methane as an energy resource, but also concern at the prospect of oceanic warming liberating the methane causing rapid global warming.
 And it's found on other planets?
And it's found on other planets?There is evidence for methane being present in several planets in the solar system, particularly Mars and Titan, Saturn's largest moon. Three groups of researchers have detected methane in spectroscopic studies of the atmosphere of Mars in 2003-4. Scientists debate whether it has a bacterial source, or if it originates in iron and magnesium containing silicate rocks, known to be capable of producing hydrogen, which could then react with carbon or its compounds forming methane.
In January 2005, the European Space Agency's Huygens probe sampled the atmosphere of Titan by GC-MS, detecting methane, but this lacked the 12C enrichment associated with living organisms, making it likely that the methane originates from geological processes somewhere in the interior. There is also liquid methane on the cold (-180°C) surface of Titan, and this has apparently carved river channels upon the surface.
 What about cows and global warming?
What about cows and global warming?Cows and other ruminants have methanogenic bacteria in their rumen which can turn digested carbon compounds into methane, emitted as flatus, some 200-300 litres of gas a day. Most of the flatus is nitrogen, but cases have been reported of cows' flatus being ignited, though not explosively. It has been suggested that nearly half methane emissions come from agriculture, with livestock and cultivation of rice prime causes (60-80 million metric tons a year each). In New Zealand, where sheep and cows cause half the nation's methane emissions, a "flatulence tax" has been suggested.

 And the termites?
And the termites?Termites use micro-organisms - either protozoa or bacteria, depending upon the termite - in the gut to help digest plant material (cellulose). An individual termite produces around 0.5 micrograms of CH4 per day; on scaling this up, world-wide emissions of up to 20 million metric tons a year from the "cathedral" nest mounds produced by the termite colonies have been estimated.
![]()
![]()