![]()
Stevioside
The ‘natural’ sweetener that’s an alternative to sugar
![]()
Paul May
University of Bristol, UK
![]()
Molecule of the Month September 2015
Also available: HTML version.
![]()

SteviosideThe ‘natural’ sweetener that’s an alternative to sugar
Paul May
Molecule of the Month September 2015
|
 |
Was it discovered by a guy named Steve, by any chance?Actually, sort of… Stevioside gets its name from the plant from which it is derived, Stevia rebaudiana, which is a shrub native to Paraguay and Brazil. The native Guarani tribes used this plant’s leaves to sweeten their tea for over 1500 years. But it wasn’t until 1899 that the plant was found and described by the Swiss botanist, Moises Santiago Bertoni, who recognised it as belonging to the same genus as an Australian weed (Stevia eupatoria) which had been named in honour of the Spanish botanist Petrus Jacobus Stevus. So, not Steve, but Stevus. Why do we need another sweetener?Well, although there are a lot of artificial sweeteners on the market, such as saccharin and aspartame, they have always been dogged by controversy over potential health issues. Despite a vast amount of scientific evidence proving that these sweeteners have no harmful effects when used normally, the internet abounds with anecdotal reports of unwanted side-effects and illnesses that may have resulted from these sweeteners – none of which have ever been verified. It didn’t help that in 1969 experiments showed that rats developed bladder cancer after being given enormous amounts of saccharin-cyclamate, leading to the FDA banning it for many years, and to the notorious ‘health warning’ on Coca Cola cans. Despite the FDA reassessing these experiments and declaring saccharin, cyclamate and aspartame as safe, and them being approved as foodstuff additives in the US and EU for over 20 years, the perception of the general public remains skeptical about their safety. Science 0, Internet rumour mill 1. |
|
Well, for a start, it’s derived from plants, so can be called a ‘natural’ product, unlike the other sweeteners which are entirely synthetic and therefore ‘artificial’.
It’s been through the usual rigorous FDA and EFSA testing procedure, and been declared safe up to a dose of 4 mg per day per kg body weight. But as usual, despite the evidence, politics and controversy aren’t far away. In 1991 the FDA suddenly labelled stevia as an ‘unsafe food product’ after receiving an ‘anonymous industry complaint’. This caused stevia to be banned from importation into the US, which led to speculation that the anonymous complainant has actually been the US manufacturers of the competing sweeteners who wanted to restrict the trade of the (mostly Chinese) stevia imports. Requests under the Freedom of Information Act failed to find out who the mysterious complainant(s) had been, because the FDA said they’d apparently deleted those names to protect its sources! It finally took until December 2008 for the FDA to approve a stevia derivative (rebaudioside A) as ‘Generally regarded as safe’ (GRAS) allowing it to be used as a food additive. But even today (2015), the FDA insist that other stevia extracts haven’t been sufficiently tested to grant them GRAS status.
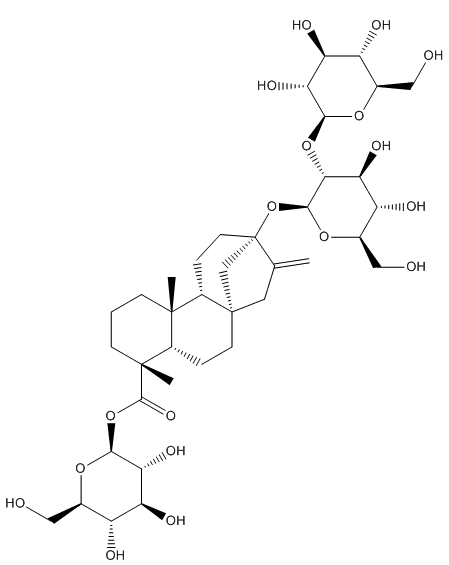
Actually, the plant contains over 40 different steviol glycosides, which are sweet-tasting molecules based around a core of steviol but with different numbers and arrangements of sugar groups (glycosides) attached to it. The glycoside with 3 sugar groups attached is called stevioside; this is the main sweet-tasting molecule extracted from stevia plants. But the glucoside with 4 glycosides attached is called rebaudioside A. This is the one that is most commonly used in commercial sweeteners because its taste most closely resembles that of sugar, although 200-300 times as sweet! It’s also heat and pH resistant, which allows it to be used in hot foodstuffs (coffee, baking, etc).
|
Steviol – the sugar groups can bind either to the topmost OH group or to the carboxylic acid group at the bottom.
 |
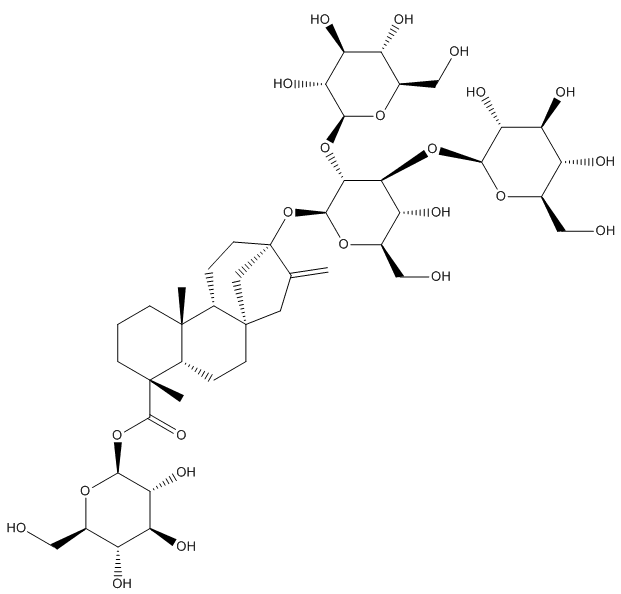 |
Stevioside – 2 sugars have bonded to the top of steviol, and one to the bottom. |
Rebaudioside A – three sugars are at the top, and one at the bottom. |
 Where can I find it?
Where can I find it?Stevia-based sweeteners come in various types depending upon which steviol glycoside they use and whether it is mixed with sugar or other sweeteners. Coca-Cola use rebaudioside A under the commercial name Rebiana. Pepsi’s version is called PureVia, while other brands include Enliten and Erylite Stevia and also Truvia although this is mixed with erythritol.
No, mixing stevia with either sugar or sugar alcohols like erythritol makes a blend that is more similar to the taste and texture of sucrose in the mouth, without adding too many calories. Erythritol is actually half as sweet as sucrose, contains almost zero calories, does not affect blood sugar levels (useful for diabetics), does not contribute to tooth decay, and causes no side-effects at normal doses. Ironically, although erythritol is naturally derived from glucose (derived from corn starch) by fermentation with a yeast, a recent lawsuit in the US has prevented it being marketed as a ‘natural’ product due to it being excessively chemically processed (i.e. chemicals such as methanol/ethanol were used as solvents!). You can never avoid politics!
|
|
One last thing, didn’t I see Stevia used in Breaking Bad?Yes, (spoiler alert for those that haven’t seen it) the character Lydia, who’s supplying the drug business in the town, often meets up with Walter or Todd to discuss how to sell and make their product. She always goes to the same café at the same time each week, orders chamomile tea with soy milk, into which we see her empty a large amount of Stevia powder sweetener. However, that consistency is her downfall, as Walter can plan how to get his revenge and poison her. In the last episode Walter mixes in some ricin into the Stevia packets, which Lydia unknowingly pours into her tea – and subsequently dies. |
![]()