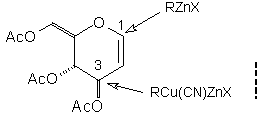
Abstract: Reaction of glycals with simple and functionalised alkyl Zn/Cu reagents in the presence of BF3.OEt2 leads to C(3)-branched carbohydrates with complete control of stereochemistry at C(3).
The reaction of organozinc nucleophiles (R2Zn or RZnX) with O-acetyl and O-benzyl-protected glycals provides a useful entry to C-glycosides (Eq. 1).1 Glycals are, however, only modest electrophiles, with the transformation shown in Eq. 1 requiring activation with an appropriate Lewis acid (BF3.OEt2 or TMSOTf). In order to extend the utility of this process we have sought to enhance the reactivity of the nucleophilic component by evaluating the corresponding Zn/Cu species.2 While these reagents retain much of the versatility associated with conventional organozinc nucleophiles, notably the ability to tolerate a spectrum of useful functional groups, a quite different pattern of reactivity towards glycals has emerged.
In earlier studies, our attempts to add (2-cyanoethyl)zinc iodide (2)3 to 3,4,6-tri-O-acetyl-D-glucal (1a) to give the C(1)-glycoside (3) were unsuccessful, a problem that has been associated with stabilisation of the organozinc species in THF solution; this reaction is successful if CH2Cl2 is used as solvent.1c However, when the corresponding Zn/Cu reagent (4)4 was employed, in the presence of up to 3 equivalents of BF3.OEt2, the C(3)adduct (5) was isolated in 91% yield (Scheme 1). Under the same conditions, the corresponding O-benzyl glucal (1b) gave adduct (6) in 51% yield. In both of these cases the new C-C bond was formed at C(3) rather than at C(1) (none of the C(1) adduct was detected) and, furthermore, C(3)-substitution occurred with net retention of configuration (see below). The scope of this process has been evaluated using both simple and synthetically more useful Zn/Cu reagents, in combination with a representative series of glycals: (1a), (1b), 3,4-di-O-acetyl-6-deoxy-L-glucal (7), 3,4-di-O-acetyl-D-xylal (8) and hepta-O-acetyl maltal (9). The results of this study are summarised in Table 1.4
Although small amounts of C(1)-adducts (cf. (3)) were isolated, generally the C(3)-branched glycal (10-16) was the major product. An exception to this was 3,4-di-O-acetyl-D-xylal (8) which underwent addition of BuCu(CN)ZnI to give approximately equal quantities of the C(3) adduct (15) and the corresponding C(1) derivative.5
The stereochemical course of the C(3) substitution appeared, based on 1H NMR correlations, to be consistent for all of the systems studied in Scheme 1 and Table 1.6 However, in the absence of an additional conformational constraint to provide more rigidity to the dihydropyran ring, stereochemical assignments by 1H NMR are less clear cut. As a consequence, the configuration at C(3) of the newly-formed C-C bond has been assigned in a more rigorous fashion. This is illustrated in Scheme 2 for two representative examples. Hydrogenation of (5) gave tetrahydropyran (17) (74% yield) and the trans relationship between H(3) and H(4) (pyran numbering) was characterised by 1H NMR.7 A similar sequence was carried out with the D-xylal-derived adduct (15) to give tetrahydropyran (18) in 62% yield.
Glycals are ambident electrophiles capable of reacting at either C(1) or C(3) with nucleophiles either directly or via the intermediacy of a Ferrier rearrangement product (a 2,3 glycal).8 Nucleophilic displacement of a C(3)-mesylate glucal derivative has been reported by Mitsunobu using either a Cu/Li or Cu/Mg reagent.9,10 Use of these more reactive organocopper reagents does limit the range of useful functional groups that can be carried within the nucleophilic component, however, Mitsunobu also observed inversion, rather than retention of configuration at C(3) of the D-glucal used in his study.

In summary, reaction of glycals, in the presence of Lewis acid activation, with Zn/Cu nucleophiles leads predominantly to the formation of C(3)-branched carbohydrate derivatives in a stereoselective fashion. This complements the regiochemical preference previously observed with conventional zinc reagents which provides an efficient and equally versatile entry to the C(1)-substituted isomers.1c
Acknowledgement. We thank EPSRC (Grant No. GR/H92142) for financial support and Professor R. F. W. Jackson for discussions.
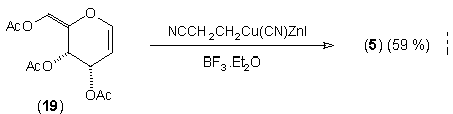
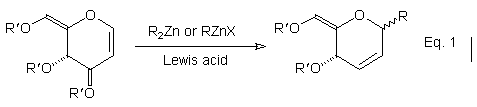
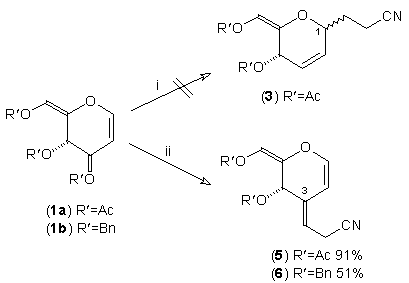
Scheme 1. Reagents: i, NCCH2CH2ZnI (2), BF3.OEt2, THF, -25°C to r.t; ii, NCCH2CH2Cu(CN)ZnI (4), BF3.OEt2,THF, -25 °C to r.t.
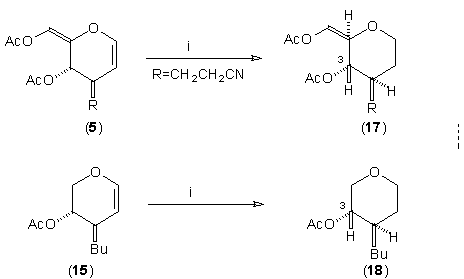
Scheme 2. Reagents: i, H2,
10% Pd on C, EtOH
| Glycal | Zn/Cu Reagent | C(3) Adduct | C(1) Adduct | % (alpha:beta) |
|---|---|---|---|---|
| (1a) | BuCu(CNZn)I | 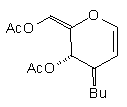
| (10) 63% | 8% (1:1.4) |
| (1a) | NC(CH2)3Cu(CN)ZnI | 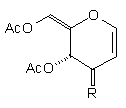
| (11) 91% R=(CH2)3CN | none |
| (1a) | Cl(CH2)4Cu(CN)ZnI | 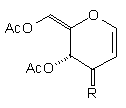
| (12) 42% R=(CH2)4Cl | 4% beta only) |
| (1a) | EtO2C(CH2)3Cu(CN)ZnI | 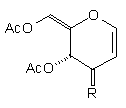
| (13) 32% R=(CH2)3CO2Et | none |
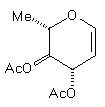 (7) | BuCu(CN)ZnI | 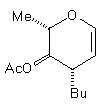
| (14) 48% | 10% (beta only) |
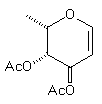 (8) | BuCu(CN)ZnI | 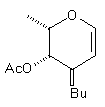
| (15) 21% | 20% (1:1.2) |
| Hepta-O-acetyl maltal (9) | NC(CH2) 2Cu(CN)ZnI | 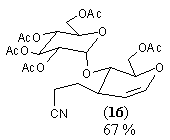 | 10% (beta only) |
| Glycals as Ambident Electrophiles Towards Organometallic Nucleophiles. Stereoselective Synthesis of C(3)-Branched Carbohydrates Simon N. Thorn and Timothy Gallagher C(3)-branched glycals, Zn/Cu nucleophiles | 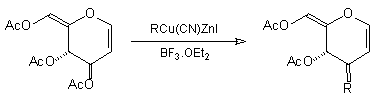
|
| Glycal Zn/Cu Reagent | C(3) Adduct | [C(1) Adduct % (alpha:beta)] |
|---|---|---|
| (1a) BuCu(CNZn)I | 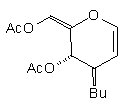
| (10) 63% [8 %; 1:1.4] |
| (1a) NC(CH2)3Cu(CN)ZnI | 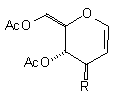 R=(CH2)3CN R=(CH2)3CN | (11) 91% [no C(1) adduct] |
| (1a) Cl(CH2)4Cu(CN)ZnI | 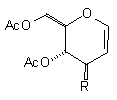 R=(CH2)4Cl R=(CH2)4Cl | (12) 42% [4% (beta only)] |
| (1a) EtO2C(CH2)3Cu(CN)ZnI | 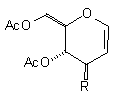 R=(CH2)3CO2Et R=(CH2)3CO2Et
| (13) 32% [no C(1) aduct] |
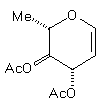 (7) (7)BuCu(CN)ZnI | 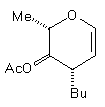
| (14) 48% [10% beta only)] |
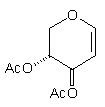 (8) BuCu(CN)ZnI (8) BuCu(CN)ZnI | 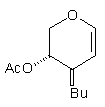
| (15) 21% [20% 1:1.2] |
| Hepta-O-acetyl maltal (9) NC(CH2) 2Cu(CN)ZnI | 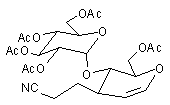 | (16) 67% [10% beta only)] |