According to its chemical structure.
According to how it is applied to materials.
There are two practical ways to classify a dye:
|
According to its chemical structure. |
|
|
According to how it is applied to materials. |
The first is of more interest to chemists, who desire to know what makes the molecule the colour it is. The second is of interest to dyers, who need to know which dye is appropriate to the material they need to dye, and the resultant colour.
This is published by the Society of Dyers and Colourists. Every commercial dye and pigment in it is given a C.I. Generic Name, which includes its application class, its hue and a number which indicates its chronological discovery2.
For example, CI Acid Violet 43 (CI 60730), CI Vat Red 41 (CI 73300),
In this method of classification, dye molecules are grouped according to shared structural groups. For example, the azo dyes, which have the general structure:
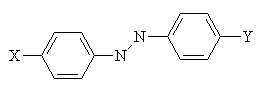
Azo dyes are the most important of the dye classes, with the largest range of colours (see Basis of Colour). All azo dyes contain at least one -N=N- group. See the Azo Dyes page for more explanation. The next most important dye class contains carbonyl functions (-C=O). This group includes anthraquinones2.
The pages that follow explain the chemistry of some classes of dyes. The dyes have been ordered as to which type of textile they dye best.