 |
Heavy WaterDeuterium Oxide, D2O
Simon Cotton
Also available: VRML, Chime Enhanced and JMol versions.
|
 |
 |
Heavy WaterDeuterium Oxide, D2O
Simon Cotton
Also available: VRML, Chime Enhanced and JMol versions.
|
 |
 Francis Aston (photo, right) found evidence for the existence of isotopes in 1913, and he published his findings in 1920. Aston was awarded the Nobel prize for Chemistry in 1922. Soon after, Rutherford suggested the existence of a heavier isotope of hydrogen, which we now call deuterium. It was first detected in 1931 by Harold Clayton Urey (see photo, below), who found that he could enrich liquid hydrogen somewhat by fractional distillation, and confirmed the cause of weak lines in the atomic spectrum of samples of hydrogen as due to the presence of small amounts of deuterium. Using electrolysis, Urey succeeded in enriching samples of water in the heavier isotope. The next step was to isolate pure heavy water. Along with his student Ronald T. MacDonald, the great American chemist Gilbert Newton Lewis (1875-1946) set to the task, using both electrolysis and fractional distillation under reduced pressure (employing a 72-feet-high distillation column). Armed with his supply of deuterium oxide, Lewis set out to investigate its properties - not just the obvious ones, like melting and boiling points - but also whether it would support life (a white mouse drank it and came back for more).
Francis Aston (photo, right) found evidence for the existence of isotopes in 1913, and he published his findings in 1920. Aston was awarded the Nobel prize for Chemistry in 1922. Soon after, Rutherford suggested the existence of a heavier isotope of hydrogen, which we now call deuterium. It was first detected in 1931 by Harold Clayton Urey (see photo, below), who found that he could enrich liquid hydrogen somewhat by fractional distillation, and confirmed the cause of weak lines in the atomic spectrum of samples of hydrogen as due to the presence of small amounts of deuterium. Using electrolysis, Urey succeeded in enriching samples of water in the heavier isotope. The next step was to isolate pure heavy water. Along with his student Ronald T. MacDonald, the great American chemist Gilbert Newton Lewis (1875-1946) set to the task, using both electrolysis and fractional distillation under reduced pressure (employing a 72-feet-high distillation column). Armed with his supply of deuterium oxide, Lewis set out to investigate its properties - not just the obvious ones, like melting and boiling points - but also whether it would support life (a white mouse drank it and came back for more).

As Paracelsus said, "Alle Ding sind Gift, und nichts ohn Gift; allein die Dosis macht, daβ ein Ding kein Gift ist", usually rendered as "the dose makes the poison". Small amounts of heavy water are not toxic, and it is thought that it would need ~30-50% of the water content of the body to be heavy water for serious poisoning to occur; this would mean the victim drinking neat heavy water for several days on end.


Left: Harold Urey Right: Gilbert Lewis
Urey was awarded the 1934 Nobel Prize in Chemistry "for his discovery of heavy hydrogen", Lewis - who had been Urey's PhD supervisor - won nothing. At this, Lewis stopped work on heavy water. Despite his work on heavy water, not to mention inventing the covalent bond (plus dot-and-cross diagrams); coming up with the concept of acids and bases as electron-pair acceptors and donors, respectively; developing a theory of electrolytes and also formulating thermodynamics for chemists (no mean feat), Lewis was never to win a Nobel prize.
There are small but measurable differences. One consequence of the D2O molecule being significantly heavier than H2O is that an ice cube made from heavy water will sink if placed in liquid H2O.
| H2O | D2O | |
| Freezing point (°C ) | 0.00 | 3.81 |
| Density of liquid (g/cm3) | 0.9999 (277 K) | 1.1056 (293 K) |
| Density of solid at m.p. (g/cm3) | 0.917 | 1.018 |
| Temperature of maximum density (°C) | 3.98 | 11.2 |
| pH (pD) (298K) | 7.00 | 7.43 |
Researchers continued to examine the properties of D2O, but obtaining large quantitites of heavy water for research was usually done by electrolysis. This required huge amounts of electricity and was then only achievable at Norsk Hydro's Ryukan plant in Norway (photo, below); they obtained really pure heavy water in early 1935. At first, demand was small, in quantities usually no more than 10 grams, but things were to change after Hahn and Strassmann's discovery in late 1938 that uranium atoms could be split using slow neutrons, releasing energy (and more neutrons). By the end of 1939, Ryukan was receiving orders from the German chemical giant I.G. Farben for up to 100 kg of D2O a month. By early 1940, a few months after the Second World War had broken out, French intelligence became aware of increased German demand for heavy water, and in early March Lieutenant Jacques Allier, a Deuxi�me Bureau agent, managed to spirit out of the country the entire Ryukan stock of heavy water, 26 five-litre containers. Shortly afterwards, another 26 containers reached Paris.

Vemork hydroelectric power station at the Rjukan waterfall in 1935.
Jean Fr�d�ric Joliot-Curie and his wife Ir�ne shared the 1935 Nobel prize for Chemistry "in recognition of their synthesis of new radioactive elements". By the outbreak of World War II, Joliot-Curie, at that time a professor at the Coll�ge de France, was working actively on nuclear fission, and since the summer of 1939 had been thinking of heavy water as a moderator in a nuclear reactor, to produce neutrons of the right velocity to induce nuclear fission. When the German invasion of France took place in May 1940, the heavy water was removed first to Clermont-Ferrand, in the Auvergne, then, after the collapse of the French Army in mid-June, Joliot-Curie oversaw its movement to Bordeaux, from where it was shipped to England aboard the SS Broompark. The world's supply of heavy water arrived in England on June 21 1940, to be stored first at Wormwood Scrubs prison, then at Windsor Castle. It was safe.
 Why did the Germans want it?
Why did the Germans want it?Like the French (and the British and the Americans), German scientists had been following developments in nuclear chemistry. The great Werner Heisenberg (who won the 1932 Nobel prize for Physics aged 31, "for the creation of quantum mechanics") was one of the leading scientists developing the German nuclear project. He had reasoned that since Germany could not separate the uranium isotopes to obtain enrichment in 235U, a reactor employing natural uranium would require a moderator of either graphite or heavy water. After the German invasion of Norway, I.G. Farben took over Norsk Hydro and the Ryukan plant.
Fed by intelligence from the Norwegian resistance movement, the British followed the increased production of heavy water at Ryukan, and decided to try to destroy the plant. On November 19 November 1942, two RAF Halifax bombers set off from Scotland, heading for Norway; each was towing a glider carrying seventeen commandos and two crew. Operation Freshman was under way. It was a disaster. One Halifax flew into a mountain, killing the crew of six; both the gliders crashed and all those who survived the crash were killed by the Gestapo. Operation Gunnerside followed, when a team of Norwegian commandos struck on the night of February 27-28 1943, causing serious damage to the plant and putting back production. In a final blow, a Norwegian resistance party sank the ferry Hydro when it was transporting heavy water to Germany on 20 February 1944. All these operations helped slow German heavy-water research. The film "Heroes of Telemark", starring Kirk Douglas and Richard Harris, is loosely based upon these events.
As it happens, German efforts were too half-hearted to bear fruit, whereas the Americans invested huge sums of money in the Manhattan project. In any case, better processes are available to obtain heavy water, such as the Girdler process, simultaneously discovered by Karl-Hermann Geib in Germany and J.S. Spevack at Columbia University. This employs an exchange reaction for which the equilibrium constant is very favourable (K~1.01):
HOH (l) + HSD (g) 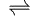 HOD (l) + HSH (g)
HOD (l) + HSH (g)
Post-war, Canada was the only country which invested heavily in plants using natural-abundance uranium which employed heavy water as the moderator.
Deuteration of compounds often assists the study of reaction mechanisms. Chemists can use D2O as a solvent for Nuclear Magnetic Resonance spectra as the different nuclear properties of deuterium means that it does not give resonances in the proton-NMR range. It can also be used to assign the signals due to labile N-H and O-H groups in compounds such as alcohols; just add a drop of heavy water to your solution of the compound in CDCl3, then the labile protons are exchanged, and the resonance due to O-H (or N-H) disappears.
R-O-H + D2O ![]() R-O-D + D-O-H
R-O-D + D-O-H
This is illustrated in the NMR spectra of menthol, below, where the OH peak at ~1.8 ppm disappears after adding D2O, showing that the H involved is free to exchange with the solvent.
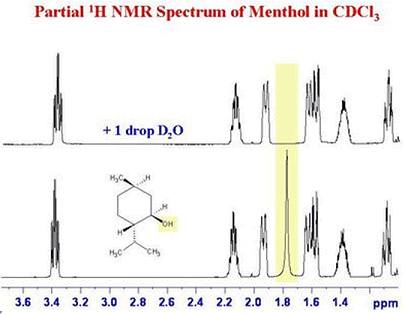
Reproduced with the kind permission of Dr. Glenn A. Facey, Department of Chemistry, University of Ottawa. Image taken from Glenn Facey's blog.
Comparing isolated H2O and D2O molecules by microwave and IR spectroscopy indicates a shortening in the bond length on passing from H2O to D2O, but only by 0.0037 Å. For an isolated H2O molecule, O-H is 0.9724 Å, and H-O-H is 104.50° whilst for an isolated D2O molecule, O-D is 0.9687 Å, and D-O-D is 104.35°. Recent research [2008] by Alan Soper of the Rutherford Appleton Laboratory (UK) and Chris Benmore from Argonne National Laboratory (USA) studying samples of liquid H2O and D2O by X-ray and neutron diffraction techniques backed up by computer simulations, has produced some surprising results. Again, the two molecules appear to have very similar, but not identical structures, but the discrepancy in bond length is much greater. They find that the O-H bond in H2O is 1.01 Å, whilst the O-D bond in D2O is 0.98 Å, shorter by 0.03 Å. On the other hand, the intermolecular bond in D2O (the "hydrogen-bond") is longer by 0.07 Å.
Soper has commented that since the proton is lighter than the deuteron, it sits higher in its potential well. "If that potential well is now perturbed by an approaching water molecule forming a hydrogen bond, then because the proton is higher in the well, it is more likely to be influenced by the approaching water molecule, drawing it away from the parent oxygen, more so than the deuteron, which is deeper in the well, and therefore less sensitive to perturbations from neighbouring water molecules." In turn, this effect may play a role in the difference between the OH and OD bond lengths.
![]()
![]()