Nobel Laureate

Bristol Chancellor
 Nobel Laureate |
We dedicate this 'Molecule of the Month to the Memory of Dorothy Crowfoot Hodgkin, Chancellor of the University of Bristol 1971-1988 |  Bristol Chancellor |
|---|
The disease, pernicious anæmia, was first described in 1821, and was invariably fatal. In 1926 Minot and Murphy, following Whipple's observation that anæmic dogs could be cured by feeding them raw liver, discovered that pernicious anæmia could also be treated by supplementing the human diet with liver. An intensive search for the "liver factor" was started. However cobalt's vital status was not proved until after World War II, in 1948, when the 'anti-pernicious anæmia factor', which became called Vitamin B12, was finally purified and isolated as crystals by Folkers and his co-workers at Merck Laboratories, and by Smith and Parker at Glaxo Laboratories. Small red crystals of Vitamin B12 were then grown by Lester Smith and given to Dorothy Hodgkin for crystal structure analysis. All that was known at this stage was that the approximate empirical formula was C61-64H84-90N14O13-14PCo.
A crystal structure on a molecule of this size and complexity had never been attempted before, it was a huge and complex task, since crystal structure determinations were not the routine tasks that they are today, and the techniques were still being developed, both the X-ray and the computer equipment were tedious and difficult to use. Thus the x-ray crystal structure which emerged from this study between 1950 the early 1960s was the first determination of a chemical formula by X-ray diffraction, and the first determination of the structure of a metalloenzyme. It was a triumph for Dorothy Hodgkin and her Oxford x-ray crystallography group, inspiring many young crystallographers, and pointing them to biochemistry as an exciting new subject for their endevours. The structure work also caused Woodward (at Harvard) and Eschenmoser (at the Swiss Federal Institute of Technology) to start synthetic work on Vitamin B12. The synthesis took 11 more years, and involved more than 90 separate reactions performed by over 100 co-workers. The sterochemical puzzles involved in the synthesis led to the Woodward-Hoffman rules. This all adds up to three Nobel prizes in chemistry and one in medicine!
Methanogens are a primitive type of bacteria, archeobacteria, capable of using methane at the end of their
electron acceptor chain, they involve a nickel corrinoid (F430) and Vitamin B12 in the catalysis of the reaction :
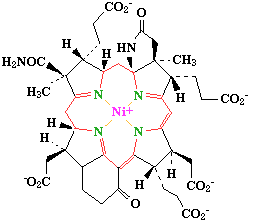
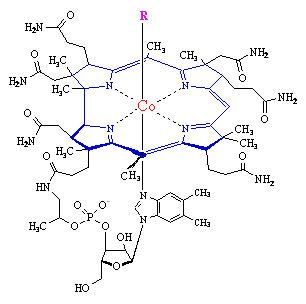 Vitamin B12
Vitamin B12 is the only known biomolecule with a stable carbon-metal bond - it is an
organometallic compound.
The core of the molecule is a corrin ring
with various attached sidegroups. The ring consists of 4 pyrrole subunits, joined on opposite sides by a
C-CH3 methylene link, on one side by a C-H methylene link, and with the two of the pyrroles joined
directly. It is thus like a porphyrin, but with one of the bridging methylene groups removed.
The nitrogen of each pyrolle is coordinated to the central cobalt atom.
The sixth ligand below the ring is a nitrogen of a 5,6-dimethylbenzimidazole. The other nitrogen is linked to a
five-carbon sugar, which in turn connects to a phosphate group, and thence back onto the corrin ring via one
of the seven amide groups attached to the periphery of the corrin ring. The base ligand thus forms a 'strap' back
onto the corrin ring.
Vitamin B12
Vitamin B12 is the only known biomolecule with a stable carbon-metal bond - it is an
organometallic compound.
The core of the molecule is a corrin ring
with various attached sidegroups. The ring consists of 4 pyrrole subunits, joined on opposite sides by a
C-CH3 methylene link, on one side by a C-H methylene link, and with the two of the pyrroles joined
directly. It is thus like a porphyrin, but with one of the bridging methylene groups removed.
The nitrogen of each pyrolle is coordinated to the central cobalt atom.
The sixth ligand below the ring is a nitrogen of a 5,6-dimethylbenzimidazole. The other nitrogen is linked to a
five-carbon sugar, which in turn connects to a phosphate group, and thence back onto the corrin ring via one
of the seven amide groups attached to the periphery of the corrin ring. The base ligand thus forms a 'strap' back
onto the corrin ring.
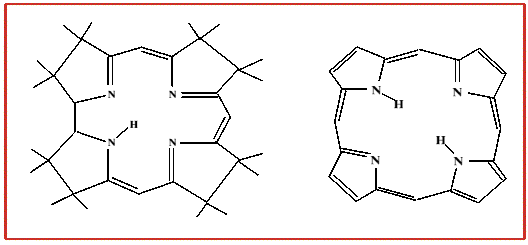 An important aspect of the corrin ring, when compared to the porphyrin,
is the relative flexibility of the corrin system, the corrin ring is also less flat when
viewed from the side than is a porphyrin ring.
This adds up to some considerable differences between the chemistry of a cobalt porphyrin
and a cobalt corrin.
An important aspect of the corrin ring, when compared to the porphyrin,
is the relative flexibility of the corrin system, the corrin ring is also less flat when
viewed from the side than is a porphyrin ring.
This adds up to some considerable differences between the chemistry of a cobalt porphyrin
and a cobalt corrin.
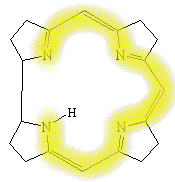 In addition, the corrin only has a conjugated chain around part of the ring system,
whereas a porphyrin is delocalised around the whole four pyrolle rings.
In addition, the corrin only has a conjugated chain around part of the ring system,
whereas a porphyrin is delocalised around the whole four pyrolle rings.
Vitamin B12 in its various forms, and in cooperation with its coenzymes and various different substrates is involved in three different forms of reaction:
The first two reactions involve a Co(II) oxidation state intermediate, the third probably involves a Co(I), and in both in its 'resting state' the cobalt is a Co(III). Central to the catalytic rôle of is the relative weakness of the cobalt-carbon bond, with a dissociation energy of around 120 kJmol-1. In several of the rearrangements, and in the reductase, EPR signals have been observed for the deoxyadenosyl radical and for a cobalt(II) corrin.
In the hydrogen migrations in all of the Vitamin B12 coenzyme-dependent rearrangement reactions, there is no exchange of the hydrogen with water protons, and the 1,2 intramolecular shifts are stereospecific for both the hydrogen atom and the exchanging group. Different reactions also proceed with either retention or inversion of the configuration.
The mechanistic picture which emerges from this is that the rearrangement involves a kind of 'reaction in a bottle', between the Vitamin B12 and the substrate, with the large coenzyme acting as the bottle. Co(II) and free radicals are very oxygen sensitive, so that the whole process must be kept anaerobic.
 is believed to be behind the
Minamata
tragedy in Japan.
is believed to be behind the
Minamata
tragedy in Japan.
The principle models used for Vitamin B12 are the alkylcobalt(III)dimethylglyoxinates,
so called alkylcobaloximes,
which have the advantages of easy preparation and simplicity.
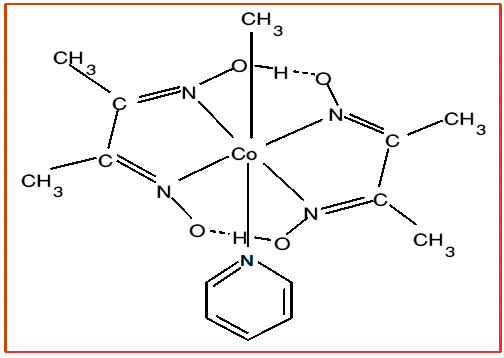 The methyl complex photolyses
readily to form ethane, and the methyl radicals can be trapped with spin-traps such as PBN. reaction with
Hg(II) forms the highly toxic MeHg(II) ion, modeling what happens when Hg(II) attacks methyl
Vitamin B12.
Of course none of these will ever provide substitutes
for Vitamin B12, but they do provide platforms on which various aspects of the mechanisms
of the Vitamin B12 catalysed reactions can be studied.
The methyl complex photolyses
readily to form ethane, and the methyl radicals can be trapped with spin-traps such as PBN. reaction with
Hg(II) forms the highly toxic MeHg(II) ion, modeling what happens when Hg(II) attacks methyl
Vitamin B12.
Of course none of these will ever provide substitutes
for Vitamin B12, but they do provide platforms on which various aspects of the mechanisms
of the Vitamin B12 catalysed reactions can be studied.