Prof Tom Simpson's Research Interests
BIO-ORGANIC AND NATURAL PRODUCT CHEMISTRY
![]()
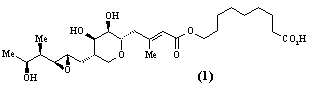
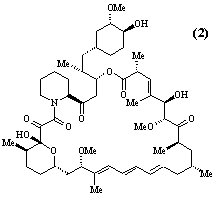
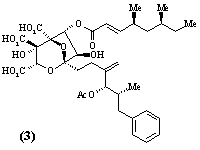
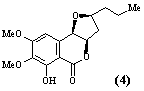
- Polyketide antibiotics. The polyketide biosynthetic pathway is responsible for the production of many antibiotics and other compounds of medicinal and agrochemical importance in a wide range of micro-organisms and higher plants. A multidisciplinary group is studying all aspects of the chemistry - isolation and structure elucidation, total synthesis and biosynthesis - of polyketide metabolites. Compounds currently being investigated include the antibiotic, pseudomonic acid (1), the immunosuppressive agent, rapamycin (2), squalestatin (3) which acts to lower blood cholesterol levels, and the insecticidal and fungicidal metabolite monocerin (4). Recent advances in genetics have enabled the biosynthetic enzymes to be isolated in sufficient quantities for mechanistic and structural studies. Projects are available which include total synthesis, synthesis of biosynthetic intermediates and enzyme substrates in isotopically labelled form, structure elucidation of novel compounds produced by genetically modified micro-organisms, and isolation and structural studies (high field NMR and molecular graphics) of biosynthetic enzymes. Much of the work in this area involves collaboration with pharmaceutical and agrochemical companies in the UK, Europe and USA, and with scientists from a wide range of disciplines, including microbiology, molecular genetics and biochemistry.
|
|
|
|
|
|
- Synthesis of biologically active natural products. Work in this area includes the total synthesis of aromatic and quinonoid compounds. Synthetic methodologies based on cycloaddition reactions of substituted pyrones and carbohydrate synthons are being developed.
- Chemistry and biosynthesis of mycotoxins. Mycotoxins are produced as a result of infestation of foodstuffs by certain fungi. Aspects being studied include synthesis of advanced intermediates for biosynthetic studies; synthesis of antigens for antibody generation; the effect of specific enzyme inhibitors on biosynthetic pathways; and chemical and biological degradation of mycotoxins.
- Novel polymers. The synthesis of novel polymers based upon substituted biphenyls and polyphenyls is being studied.
Relevant
References:
1.
T.J. Simpson,
"Applications of Multinuclear NMR to Structural and Biosynthetic Studies
of Polyketide Microbial Metabolites", Chem. Soc. Review, 1987, 16,
123-160.
2.
J.A. O'Neill,
T.J. Simpson and C.L. Willis, "Biosynthesis of Colletodiol and Related
Polyketide Macrodiolides in Cytospora sp. ATCC 20502: Synthesis and
Metabolism of Advanced Intermediates", J. Chem. Soc., Chem. Comm.,
1993, 738.
3.
J.A. O'Neill,
T.J. Simpson and C.L. Willis, "Structures of Bartanol and Iso-bartanol,
Novel Macrodiolide Metabolites from Cytospora sp ATCC 20502", J.
Chem. Soc., Perkin Trans. 1, 1994, 2493-2497
4.
T.J. Simpson,
"Polyketide Biosynthesis", Chem. Ind., 1995, 407-411; J.A.
O'Neill, S.D. Lindell, T.J. Simpson and C.L. Willis, "Enantioselective
Synthesis of the 13-Membered Macrodiolide Bartanol", J. Chem. Soc.,
Perkin Trans. 1, 1996, 632-644.
5.
D.S.J. McKeown,
C. McNicholas, T.J. Simpson and N.J. Willett, "Biosynthesis of
Norsolorinic Acid and Averufin: Substrate Specificity of Norsolorinic Acid
Synthase", J. Chem. Soc., Chem. Commun., 1996, 301-302.
6.
C. McNicholas,
T.J. Simpson and N.J. Willett, "Enantioselective Synthesis of Fusarentin
Methyl Ethers: Insecticidal Metabolites of Fusarium larvarum",
Tetrahedron Lett., 1996, 37, 8053-8056.
7.
M.P. Crump, J.
Crosby, C.E. Dempsey, J.A. Parkinson, M. Murray, D.A. Hopwood and T.J. Simpson,
"The Solution Structure of the Actinorhodin Polyketide Synthase Acyl
Carrier Protein from Streptomyces coelicolor A3(2)",
Biochemistry, 1997, 36, 6000-6008.
- T.J. Simpson, R.W. Smith, S.M.Westaway, C.L. Willis, A.D. Buss,
R.J.P. Cannell, M.J. Dawson and B.A.M. Rudd, "Enantioselective
Synthesis of a Putative Hexaketide Intermediate in the Biosynthesis of the
Squalestatins", Tetrahedron Lett., 1997, 38,
5367-5370.
![]() Back to School of
Chemistry Home Page.
Back to School of
Chemistry Home Page.